DNA/RNA Drug Purity Testing Service
MtoZ Biolabs provides a specialized DNA/RNA Drug Purity Testing Service to support the development and quality control of nucleic acid therapeutics. This service ensures accurate determination of purity levels, offering reliable data that underpins safety, efficacy, and stability across the drug lifecycle.
DNA and RNA therapeutics, such as antisense oligonucleotides, siRNA, mRNA, aptamers, and plasmid-derived products, have revolutionized modern medicine. These modalities enable targeted gene silencing, protein expression, or modulation of cellular pathways, making them powerful tools for treating cancer, genetic disorders, infectious diseases, and rare conditions. However, their inherent complexity and chemical modifications demand rigorous purity testing. Even small amounts of impurities, such as truncated sequences, modified nucleotides, or degradation products, can compromise therapeutic performance or trigger adverse immune responses.
DNA/RNA drug purity testing refers to the systematic evaluation of the proportion of intact nucleic acid molecules within a preparation, separated from impurities. Impurities may arise during synthesis, formulation, storage, or transport. Comprehensive purity testing therefore plays a pivotal role in process optimization, regulatory submission, and clinical safety assurance.
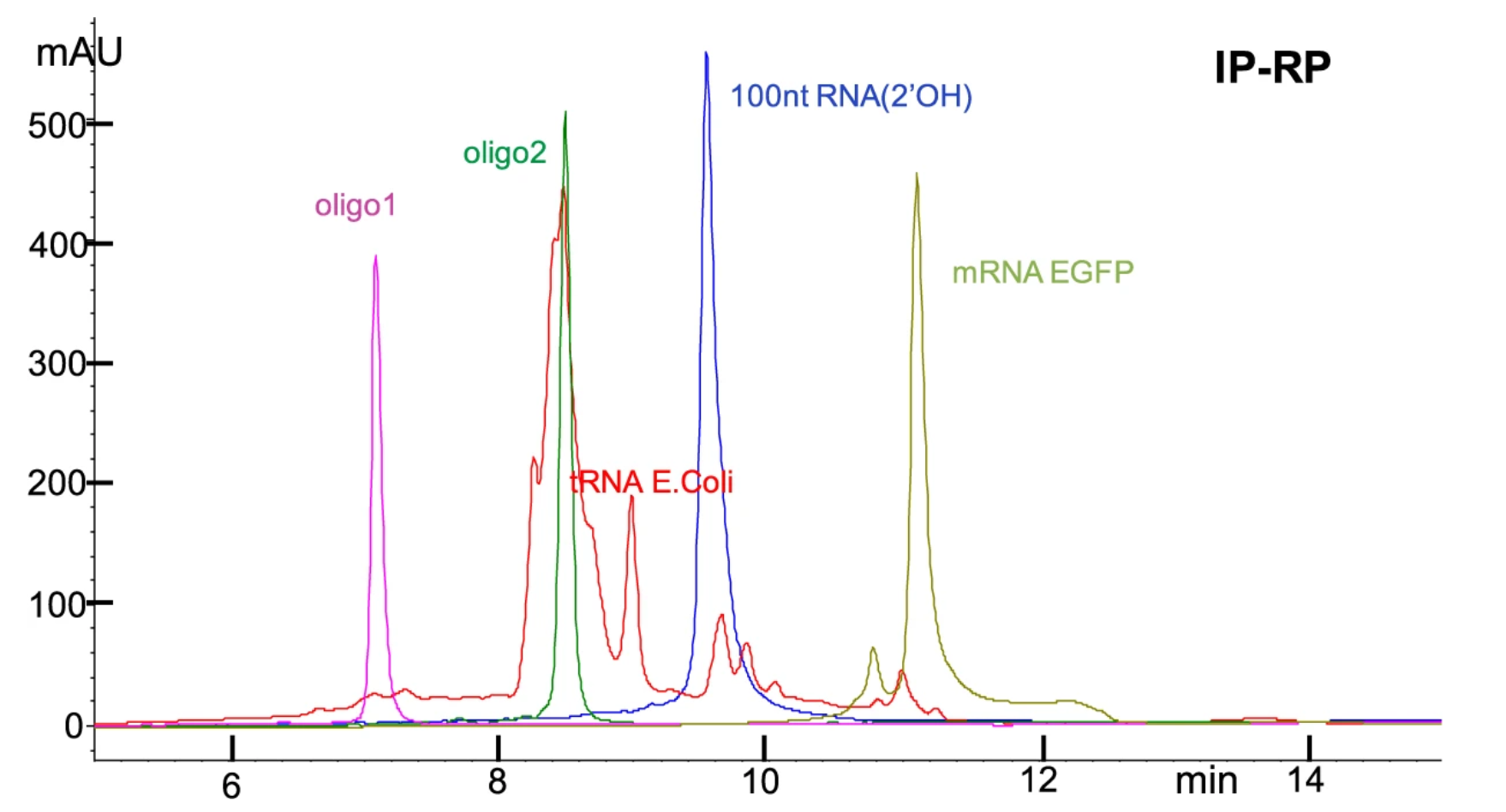
Figure 1. HPLC Profiles for Purity Evaluation of Man-Made Single-Stranded RNAs
Service at MtoZ Biolabs
MtoZ Biolabs' DNA/RNA Drug Purity Testing Services help meet the highest standards of quality and safety for nucleic acid therapeutics. Our analytical solutions are designed to detect, quantify, and characterize impurities while delivering accurate purity profiles tailored to client needs.
Our advanced analytical methods include:
🔸Liquid Chromatography coupled with Mass Spectrometry (LC/MS)
Enables high-resolution separation and precise mass identification of intact DNA/RNA molecules, impurities, and degradation products. This approach provides detailed molecular characterization and reliable quantification.
🔸Capillary Gel Electrophoresis (CGE)
Offers sensitive and high-resolution separation of nucleic acid species based on size and charge, enabling detection of truncated products and assessment of overall purity.
🔸UV Absorption Spectroscopy
Provides rapid evaluation of nucleic acid concentration and purity, serving as a complementary method to verify sample integrity before advanced analyses.
By integrating these complementary methods, MtoZ Biolabs ensures robust, reproducible, and comprehensive assessment of DNA/RNA drug purity. Our tailored workflows are adaptable to a wide range of drug types, including oligonucleotides, siRNA, antisense molecules, mRNA, and plasmid DNA fragments.
Analysis Workflow
Service Advantages
☑️Advanced Technology: Access to state-of-the-art HPLC, CGE, MS, MALDI-TOF, and PAGE platforms.
☑️Expertise: Experienced scientists with strong backgrounds in nucleic acid chemistry and pharmaceutical analytics.
☑️Customized Solutions: Tailored workflows designed to meet specific project needs and regulatory expectations.
☑️Efficiency: Reliable, fast, and reproducible results with streamlined processes.
☑️Comprehensive Support: From method planning to data interpretation, we provide end-to-end project assistance.
Applications
Our DNA/RNA Drug Purity Testing Service supports multiple stages of drug development and production:
1. Drug Discovery and Development
Purity testing ensures candidate DNA/RNA drugs are free from unwanted by-products, enabling reliable evaluation of therapeutic potential.
2. Manufacturing Quality Control
Routine analysis monitors production consistency and detects impurities that could compromise safety or efficacy.
3. Regulatory Compliance
Provides validated purity data packages required by global health authorities to support approval processes.
4. Stability Studies
Identifies degradation patterns under stress and storage conditions, guiding formulation design and shelf-life determination.
5. Batch-to-Batch Consistency
Compares purity profiles across production lots, ensuring reproducibility and minimizing variability.
6. Clinical Research
Verifies the integrity of nucleic acid drugs used in trials, safeguarding patient safety and study reliability.
Sample Submission Suggestions
1. Sample Types
Acceptable samples include DNA drugs, RNA drugs, oligonucleotides (modified or unmodified), and aptamers.
2. Storage and Transportation
Samples should be stored at low temperature (-20°C or below), and shipment on dry ice is recommended.
It is recommended to contact the MtoZ Biolabs technical team prior to sample submission to obtain detailed and tailored guidelines for sample preparation and submission.
Deliverables
1. Detailed Experimental Protocols
2. Chromatograms and Electropherograms
3. Mass Spectra and Sequence Confirmation Results
4. Purity Percentages and Impurity Profiles
5. Data Interpretation and Conclusions
6. Raw Data Files
MtoZ Biolabs is committed to providing reliable, comprehensive, and high-quality analytical solutions to support every stage of DNA/RNA drug development. Free project evaluation, welcome to learn more details!
Related Services
DNA/RNA Drug Molecular Weight Confirmation Service
How to order?







