Research on Media Development and Optimization
Culture media are essential mixtures designed to support the growth and metabolism of microorganisms, cells, or tissues. These media provide crucial nutrients and environmental conditions, typically including water, carbon and nitrogen sources, inorganic salts, vitamins, and growth factors. The pH of the media is carefully adjusted to the optimal levels for specific organisms or cells.
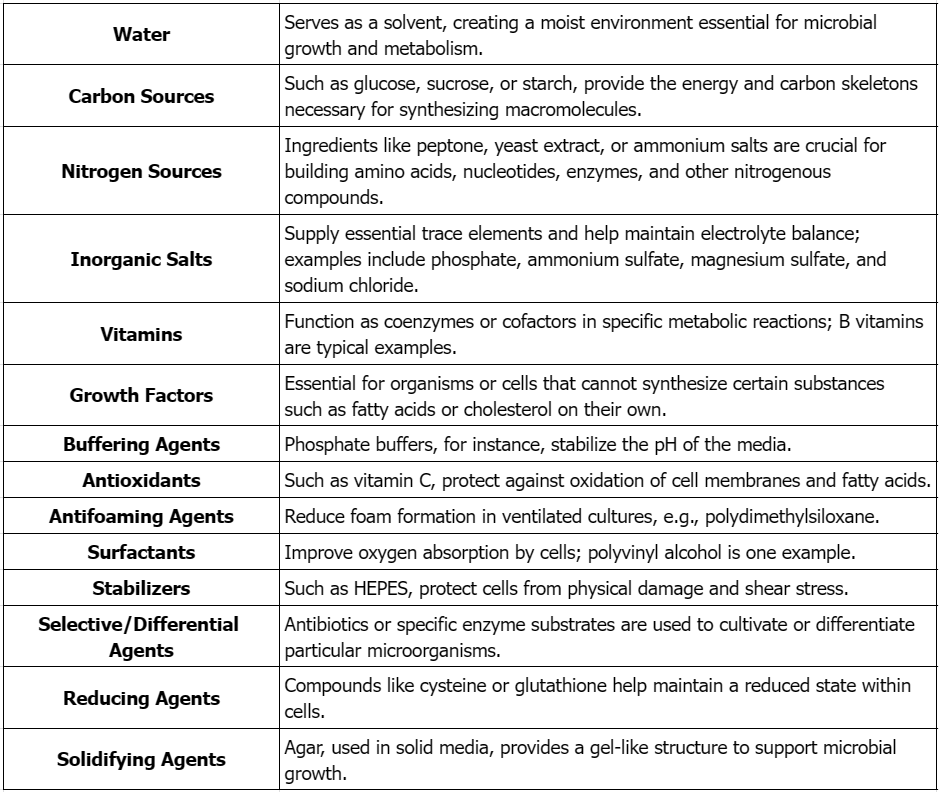
1. Main Components and Functions of Culture Media
2. Media Classification
(1) Natural Media: Typically made from natural substances like meat broth or yeast extract, used for general culturing.
(2) Synthetic Media: Composed of precisely measured chemical components, used for research and exact control of experimental conditions.
(3) Solid Media: Contains solidifying agents like agar for microbial isolation and quantification.
(4) Liquid Media: Predominantly liquid, suitable for microorganisms or cells that need to be grown in suspension.
(5) Selective Media: Contains inhibitors or enhancers for growing specific microorganisms.
(6) Differential Media: Identifies microbes based on their metabolic products.
(7) Enriched Media: Nutrient-rich for cultivating nutritionally demanding microorganisms.
(8) Minimal Media: Minimal nutrients for basic metabolic studies.
(9) Maintenance Media: Stabilizes microbial growth and prevents mutation for long-term preservation.
(10) Anaerobic Media: Supports the growth of strictly anaerobic microorganisms under oxygen-free conditions.
(11) Tissue Culture Media: Supports the growth and division of animal cells.
(12) Fermentation Media: Optimized for high-yield, efficient large-scale industrial fermentation.
(13) Cell Culture Media: Simulates in vivo conditions for mammalian cell culture.
(14) Special Purpose Media: Designed for specific microbial types or experimental aims.
3. Optimization Steps
(1) Media Selection: Perform tests with different media to understand their impacts.
(2) Amino Acid Analysis: Best conducted with cooperation between media providers and pharmaceutical companies or by certified labs.
(3) Data Analysis: Calculate amino acid compositions and assess their impact using analysis of variance (ANOVA).
(4) Verification: Optimize and test amino acids identified as critical to refine the media formula.
Testing Content Required by Policies and Regulations
Class II Special Controls Guidance Document: Tissue Culture Media for Human ex vivo Tissue and Cell Culture Processing Applications; Final Guidance for Industry and Reviewers
1. Product Description
(1) A detailed description of the base culture media and a list of all potential additives or supplements for customizing the tissue culture media, including the expected range for each. Purity levels, such as USP grade, are specified for each component. If components are not USP grade, provide adequate justification to demonstrate that the chemicals possess sufficient purity for use in this product.
(2) A general explanation of the purpose of each chemical component, (e.g., prevention of swelling, metabolic support.)
(3) The identity of all packaging materials.
2. Performance Testing
(1) Evidence should be submitted to demonstrate the lack of toxicity and the performance for maintenance of cell function.
3. Sterility and Endotoxin Testing
Details of the sterilization method and level of sterility assurance are provided, along with proof of sterility, freedom from pyrogens, and, if applicable, absence of foreign agents.
The Special 510(k) Program Guidance for Industry and Food and Drug Administration Staff
1. Product Description
A complete solution description is needed, including the basic culture media and any additives or supplements that might be included.
2. Performance Testing
Evidence should be submitted to prove that lacks toxicity and maintains cell function.
3. Sterility and Endotoxin Testing
A description of the sterilization method and sterility assurance level is required.
4. Labeling
The label must include the prescription use statement found in 21 CFR 801.109(b)(1).
5. Risk Assessment
Identified health risks associated with the use of the device, including toxicity to cells or tissues, microbial contamination, inadequate support for cells or tissues, and transfer of chemical impurities.
Media Optimization Evaluation Solutions
Optimizing the ratios of nutritional components in culture media is an intricate process that typically involves the following well-defined steps:
1. Literature Review
Conduct a thorough review of existing scientific literature to understand the formulations of media used in similar studies for specific types of cells or microbes.
2. Basic Media Selection
Select a foundational media that suits the target cells or microbes, serving as the initial point of reference.
3. Nutrient Analysis
Analyze the metabolic pathways and nutritional requirements of the target organism to pinpoint essential nutrients, such as carbon and nitrogen sources, vitamins, and minerals.
4. Small-Scale Experimental Testing
Evaluate different nutrient ratios in small-scale experiments to observe their effects on cell growth or product expression.
5. Experimental Design
Employ statistical methods, including ANOVA and orthogonal designs, to systematically alter and assess various nutrient ratios.
6. Response Surface Methodology (RSM)
Utilize RSM in experimental design, a sophisticated mathematical and statistical approach that assesses the effects and interactions of multiple variables.
7. Stepwise Optimization
Gradually adjust component ratios to monitor their effects on cell growth or product expression, methodically working towards the optimal balance.
8. Bioassays
Implement appropriate bioassay techniques, such as cell counting, growth curves, and analysis of metabolic products, to evaluate the efficacy of different media formulations.
9. Cost-Effectiveness Analysis
Assess the cost-effectiveness of media formulations while ensuring their culturing efficacy, selecting the most economically viable option.
10. Stability and Consistency Testing
Evaluate the stability and batch-to-batch consistency of the optimized media.
11. Scale-Up Considerations
Address potential issues during the transition from laboratory scale to industrial production, ensuring that the media formulation consistently performs well at all scales.
12. Safety and Regulatory Compliance
Verify that the media components and their use comply with biosafety and industry regulations.
13. Feedback and Iteration
Continuously refine the media formulation based on experimental outcomes, integrating feedback for iterative optimization.
14. Expert Consultation
During optimization, seek insights and recommendations from experts within the field.
15. Software Simulation
Leverage bioprocess simulation software to predict the impacts of various media formulations on cellular metabolism and growth.
Service Advantages
1. Professional and Robust Services Covering a Wide Range of Media Development and Optimization Research
2. High Cost-Effectiveness, Short Testing Cycles, and Reliable, Comprehensive Test Reports
3. Equipped with a Variety of High-Resolution MS and other Major Equipment
Case Study
1. Challenges of Developing Cell Culture Media Using Machine Learning
Microbial and mammalian cells are widely used in the food, pharmaceutical, and medical industries. As a key technology in cell culture engineering, developing or optimizing media is crucial for enhancing cell culture performance. The methods for media optimization have significantly advanced, such as the one-factor-at-a-time (OFAT) and RSM. This review summarizes the emerging machine learning (ML) technologies in cell culture engineering, combined with high-throughput experimental techniques to develop efficient media. It discusses commonly used ML algorithms and successful applications of ML in media optimization. The paper highlights the advantages of ML-assisted media development and guides the selection of media optimization methods suitable for various cell culture purposes.
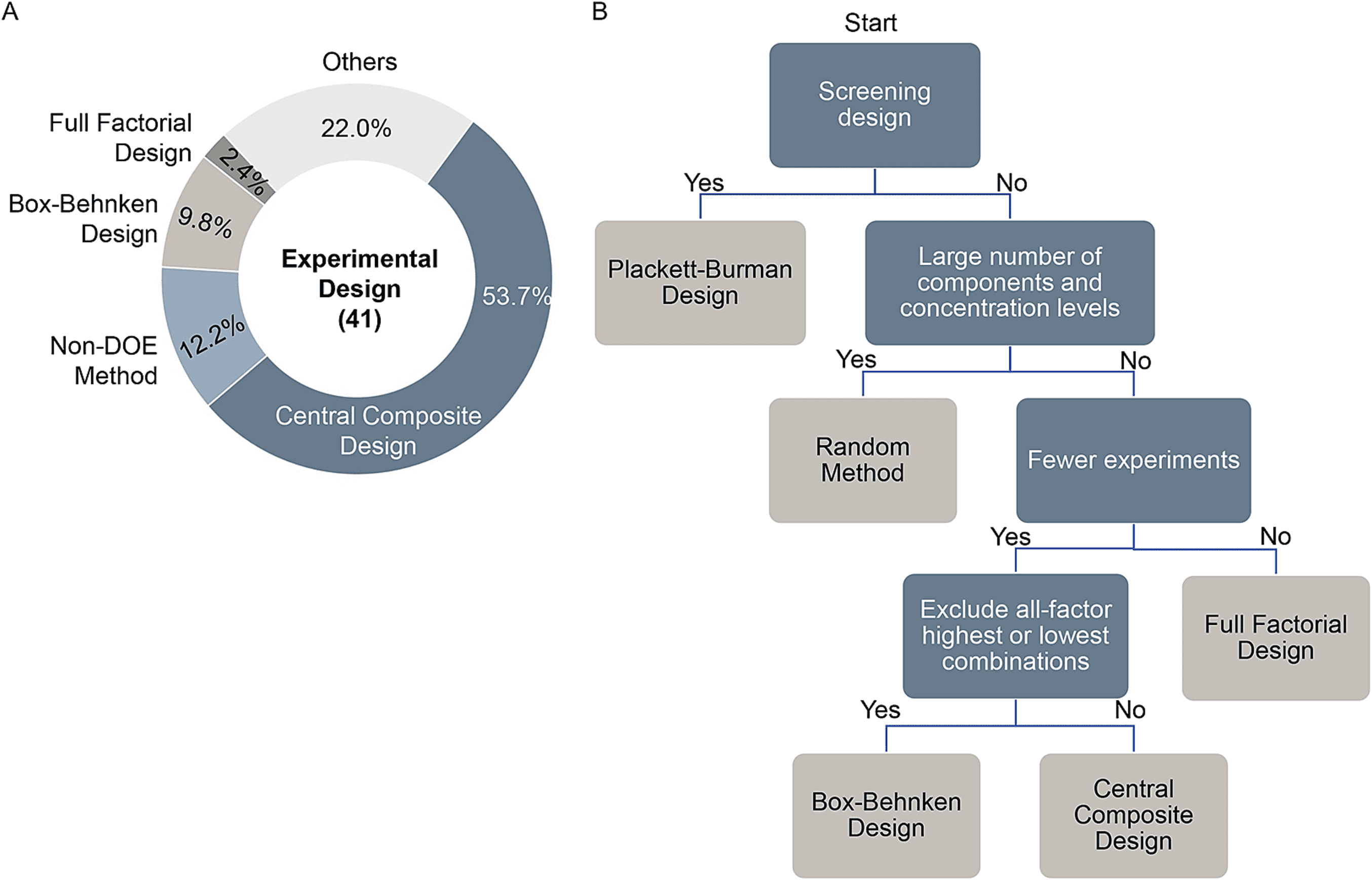
Figure 1. Experimental Design for Media Optimization [1]
2. Adopting Active Learning in Mammalian Cell Culture Media Optimization
Media optimization is a critical step in biopharmaceutical and regenerative medicine cell culture processes; however, it remains challenging due to the complexity of both the media and the cells. Research has addressed this issue by adopting active learning. Specifically, machine learning was introduced into cell culture experiments to optimize the media. The HeLa-S3 cell line and gradient-boosting decision tree algorithm were used to identify optimized media in a pilot study. Training data was collected by culturing cells in various media combinations. The cellular NAD(P)H abundance, represented as A450, was used to indicate the goodness of culture media In active learning, both a regular mode and a time-saving mode were developed using 168-hour and 96-hour culture data, respectively. Both modes successfully fine-tuned 29 components to produce media for improved cell culture. Interestingly, these two modes provided different predictions for the concentrations of vitamins and amino acids, with fetal bovine serum (FBS) concentrations typically predicted to significantly decrease compared to commercial media. Additionally, active learning-assisted media optimization significantly enhanced cell NAD(P)H concentrations, an active chemical substance with a constant abundance in living cells. The study demonstrated the efficiency and practicality of active learning in media optimization, providing valuable information for the application of machine learning techniques in cell biology experiments.
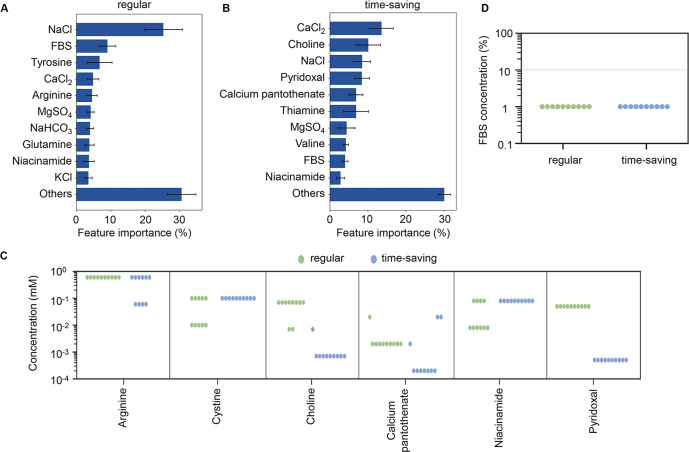
Figure 2. Contributions and Composition of Optimized Media Components [2]
3. Assessment of Cultured Media for Desert Moss Crust by Physiological Responses
The physiological responses of desert moss crusts under four artificial media (Beneck, Part, BG11, and Hogland) were studied to evaluate the functionality of the media during different culturing periods. Results showed that malondialdehyde (MDA) levels peaked at 11 days, while chlorophyll-a, soluble protein, and soluble sugar levels were at their lowest. Over time, MDA and soluble protein levels decreased more rapidly in Hogland, while chlorophyll-a and soluble sugar levels increased. At the end of the culturing period, chlorophyll-a and soluble sugar levels were highest in Hogland, while MDA and soluble protein levels were lowest. The results indicated that Hogland media promotes the growth of desert moss crusts. There is a general expectation that the selected artificial media could be more broadly and extensively applied in the field cultivation of desert biological crusts.
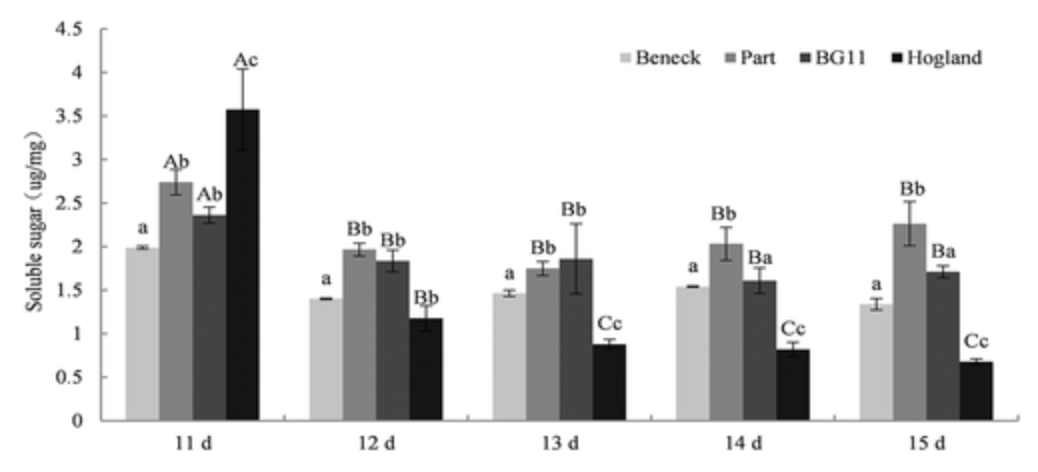
Figure 3. Soluble Sugar Content in Desert Moss Crusts Across Four Artificial Media [3]
References
[1] Hashizume T, Ying BW. Challenges in developing cell culture media using machine learning. Biotechnol Adv. 2024 Jan-Feb;70:108293. doi: 10.1016/j.biotechadv.2023.108293. Epub 2023 Nov 19. PMID: 37984683.
[2] Hashizume T, Ozawa Y, Ying BW. Employing active learning in the optimization of culture medium for mammalian cells. NPJ Syst Biol Appl. 2023 May 30;9(1):20. doi: 10.1038/s41540-023-00284-7. PMID: 37253825; PMCID: PMC10229643.
[3] Tao L, Ren H, Ren J. Assessment of cultured media for desert moss crust by physiological responses. J Basic Microbiol. 2021 Feb;61(2):157-164. doi: 10.1002/jobm.202000665. Epub 2021 Jan 4. PMID: 33393125.
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
16S rRNA Sequencing Bacterial Identification Service
How to order?







