UGT Inhibition Assays Service
MtoZ Biolabs provides comprehensive UGT Inhibition Assays Service to support drug discovery, ADME profiling, and drug-drug interaction risk assessment. Our service combines validated in vitro enzymatic systems, high-resolution analytical platforms, and expert data interpretation, offering researchers highly reliable inhibition characterization for a wide range of small-molecule and peptide drug candidates.
What Is UGT?
UDP-glucuronosyltransferases (UGTs) are essential phase II metabolic enzymes responsible for glucuronidation, a major detoxification process that facilitates the elimination of xenobiotics, endogenous compounds, and therapeutic agents. Inhibition of UGT isoforms can significantly alter drug clearance, increase systemic exposure, and lead to clinically relevant drug-drug interactions. As a result, early evaluation of UGT inhibition is critical for predicting metabolic liabilities, guiding lead optimization, and ensuring safety in downstream preclinical and clinical studies.
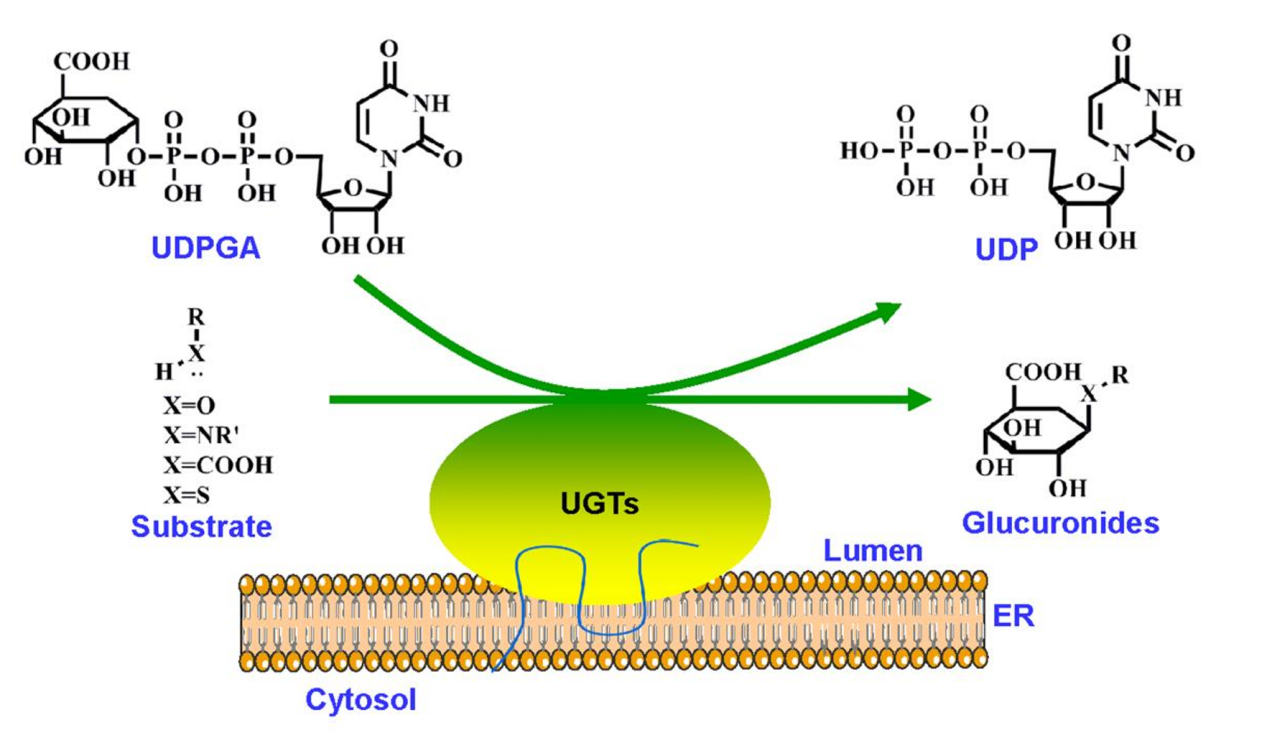
Lv, X. et al. Biotechnol J. 2019.
Figure 1. A Schematic Presentation of UGT-Catalysed Glucuronidation Reactions
UGT inhibition assessment provides mechanistic insights into how candidate molecules modulate glucuronidation pathways. By quantifying inhibitory potency and determining kinetic properties such as IC values and inhibition mode, researchers can identify whether a compound may interfere with major UGT isoforms. These data play an essential role in regulatory submissions, risk evaluation, and rational modification of chemical structures.
UGT Inhibition Assays Service at MtoZ Biolabs
MtoZ Biolabs offers an end-to-end UGT Inhibition Assays Service built upon validated in vitro systems, highly reproducible experimental workflows, and advanced analytical capabilities. Our service enables detailed evaluation of candidate compounds across multiple UGT isoforms including UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, and UGT2B15.
Our service framework includes:
💠Inhibition testing against human liver microsomes, recombinant UGT isoforms, and hepatocyte preparations.
💠Quantitative LC-MS/MS analysis of glucuronide formation to measure inhibition potency.
💠Determination of inhibition constants, including IC50 and Ki, to characterize inhibitory strength under defined kinetic conditions.
💠Assessment of inhibition mechanisms, covering competitive, non-competitive, uncompetitive, and mixed inhibition models to clarify how test compounds modulate UGT-mediated metabolism.
💠Kinetic property evaluation to support mechanistic interpretation and refine UGT interaction risk profiles.
Why Choose MtoZ Biolabs
✔️Extensive experience with a full panel of clinically relevant UGT isoforms.
✔️High-precision LC-MS platforms enabling reliable quantification.
✔️Tailored assay formats to meet discovery, optimization, or regulatory needs.
✔️Transparent communication and customizable project design.
✔️Rapid turnaround and strict quality control.
Applications of UGT Inhibition Assays Service
1. Drug-drug interaction risk assessment
UGT inhibition data support early identification of metabolic interference between candidate compounds and co-administered drugs, helping predict clinically relevant DDIs.
2. Metabolic pathway elucidation
Inhibition profiling across UGT isoforms aids in defining major metabolic routes and determining whether glucuronidation is a primary clearance mechanism.
3. Lead optimization in drug discovery
IC50, Ki, and kinetic data allow structure-activity relationship analysis, guiding chemical modification to minimize UGT inhibition liabilities.
4. Preclinical safety evaluation
UGT inhibition testing helps identify compounds with potential metabolic bottlenecks or toxic metabolite accumulation during preclinical development.
FAQ
Q1: What types of samples are suitable?
We accept a wide range of sample types for UGT inhibition assessment.
1. Pure compounds or peptide candidates intended for metabolic evaluation
2. Biological samples including cells, tissues, and biofluids when used as sources of metabolic components or when assessing endogenous inhibitory effects
Q2: How should I prepare my samples?
1. Ensure samples are freshly prepared or snap-frozen.
2. Avoid detergents, salts, and reducing agents that may interfere with LC-MS/MS analysis.
3. Store samples at –80 °C before shipment.
4. Transport samples on dry ice to maintain stability during delivery.
For more information, please refer to Sample Submission Guidelines for Proteomics and Sample Submission Guidelines for Metabolomics.
Q3: What is the service general workflow?
Q4: What data formats are provided?
We deliver results in multiple professional formats suitable for downstream interpretation and regulatory use.
1. PDF reports summarizing experimental design, kinetic findings, and inhibition interpretation
2. Raw and processed LC-MS/MS data in formats such as RAW, mzML, or mzXML
3. Quantitative results and kinetic parameters including IC50 and Ki values in Excel or CSV format
4. Chromatograms and spectra in high-resolution PNG or TIFF formats
5. Additional data formats can be provided upon request to support specific analysis or publication requirements.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs is committed to delivering high-quality UGT inhibition assessment to support drug discovery and safety evaluation.
Contact us to discuss your research requirements and receive a customized plan tailored to your research goals.
How to order?







