UGT Induction Assays Service
-
Pooled human hepatocytes for population-based responses
-
Single donor hepatocytes for donor specific evaluation
-
Identification of inducers that accelerate drug clearance
-
Prediction of exposure changes in clinical settings
-
Comparison of candidate molecules within a chemical series
-
Assessment of induction risk for coadministered compounds
-
Support for regulatory submissions requiring induction data
-
Investigation of metabolic activation routes
-
Evaluation of adaptive responses to chemical stress
-
Exploration of nuclear receptor mediated regulation
-
Characterization of phase II enzyme networks
-
Understanding metabolic pathways for new chemical entities
-
Identification of metabolites and glucuronide profiles
MtoZ Biolabs provides UGT Induction Assays Service to support researchers and pharmaceutical developers in evaluating how test compounds influence the induction of uridine diphosphate glucuronosyltransferase enzymes. This service focuses on assessing transcriptional and functional changes in UGT activity and provides quantitative data essential for understanding drug metabolism, potential interactions, and overall pharmacokinetic behavior.
What Is UGT Induction?
UDP-glucuronosyltransferases are a major family of phase II drug metabolizing enzymes responsible for glucuronidation reactions. These enzymes catalyze the conjugation of endogenous and exogenous compounds with glucuronic acid, promoting solubility and facilitating excretion. UGTs participate in the metabolism of a broad range of small molecules including therapeutic agents, steroids, bilirubin, bile acids, xenobiotics, and environmental chemicals.
UGT induction refers to the upregulation of UGT gene expression or enzyme activity in response to chemical exposure. Induction increases metabolic clearance and may reduce drug exposure, alter pharmacokinetic profiles, or interact with coadministered compounds. Regulatory agencies consider UGT induction an important determinant of drug safety, potential drug drug interactions, and metabolic liabilities. Therefore, reliable in vitro induction studies are required during drug development to predict in vivo outcomes.
UGT induction assays are designed to quantify changes in transcriptional levels, protein abundance, and catalytic activity. These assays help identify compounds that activate nuclear receptors such as PXR, CAR, or AhR, which are known regulators of both phase I and phase II enzymes. By assessing the molecular response in hepatocytes, developers can make informed decisions regarding clinical dosing strategies, metabolic risk, and regulatory compliance.
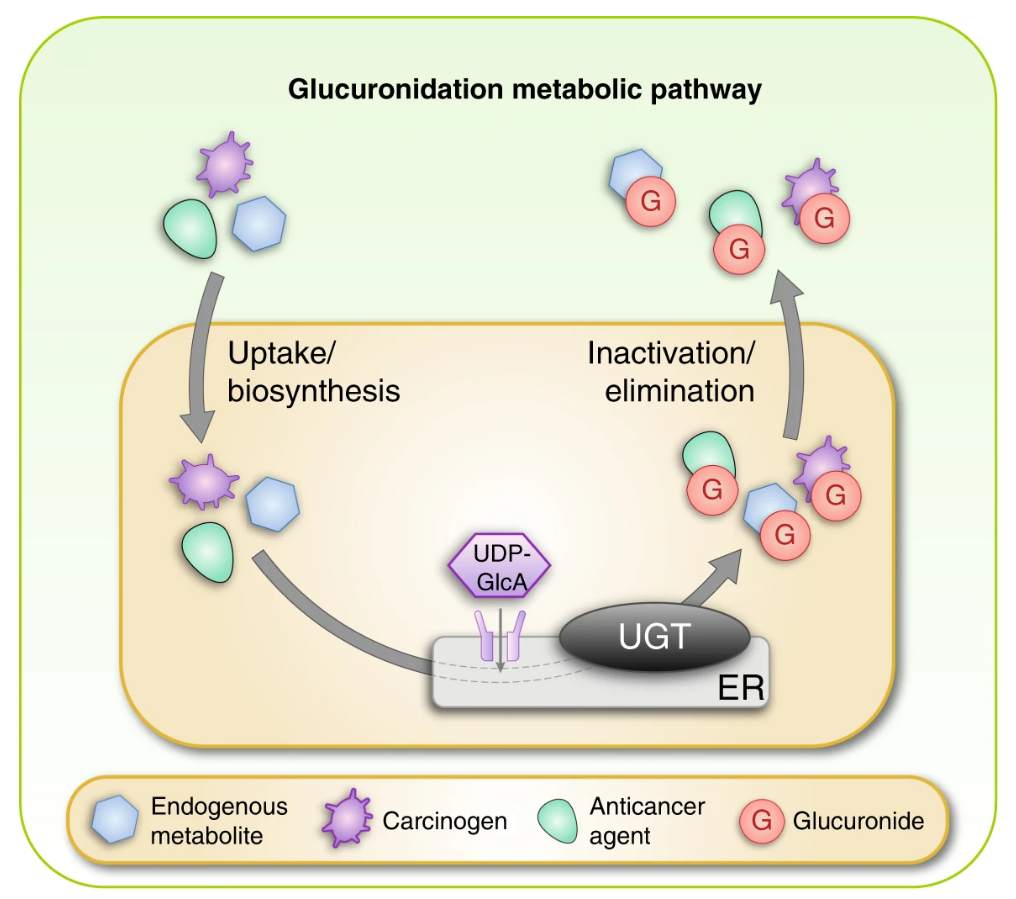
Allain, E. P. et al. Br J Cancer. 2020.
Figure 1. Schematic Overview of the Glucuronidation Reaction Catalysed by UGT Enzymes
UGT Induction Assays Service at MtoZ Biolabs
Our UGT Induction Assays Services combine state of the art hepatocyte models with quantitative molecular and enzymatic measurements, enabling a comprehensive assessment of induction potential across multiple UGT isoforms including UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, and UGT2B15.
1. UGT Induction Profiling
We conduct dedicated induction assays that evaluate the response of UGT enzymes to test compounds across multiple concentrations and time points. These assays capture early transcriptional changes as well as downstream functional effects, ensuring complete representation of the induction cascade.
2. Quantitative mRNA Measurement
To determine whether a compound upregulates UGT genes, we measure changes in mRNA expression using real time quantitative PCR. This approach provides sensitive and reproducible fold change analysis, enabling detection of both subtle and robust induction events.
3. Enzyme Activity Evaluation
Functional induction is assessed by analyzing glucuronidation activity using LC-MS-based detection. Probe substrates are incubated with hepatocytes following treatment, and metabolite formation is quantified to determine whether the test compound increases catalytic turnover. This endpoint directly reflects metabolic capacity and supports drug drug interaction predictions.
4. High Quality Hepatocyte Systems
Our assays are available in multiple biological models, including:
These systems maintain key metabolic pathways and nuclear receptor signaling required for accurate induction assessments.
5. Multi Endpoint Analysis
To support comprehensive interpretation, our service includes both gene level and enzyme activity endpoints. By integrating transcriptional and functional data, MtoZ Biolabs provides a coherent and mechanistic understanding of the induction profile for each test compound.
6. Advanced Analytical Technologies
All induction studies leverage optimized qPCR platforms for gene expression quantification and LC-MS/MS instrumentation for precise metabolite measurement. These technologies ensure high sensitivity, broad dynamic range, and robust reproducibility across experiments.
Why Choose MtoZ Biolabs
✔️High Technical Sensitivity
We use optimized qPCR protocols and advanced LC-MS platforms that ensure precise quantification even at low induction levels.
✔️Experienced Scientific Team
Our staff have extensive experience in drug metabolism studies, hepatocyte culture, and induction assay design.
✔️Regulatory Ready Data
All data sets are generated with traceable procedures and structured documentation suitable for regulatory review.
✔️Customized Experimental Design
Each project is tailored to the compound class, intended application, and data requirements.
✔️Fast Turnaround and Clear Communication
We maintain efficient workflows and provide continuous updates throughout the study.
Applications of UGT Induction Assays Service
UGT Induction Assays Service is applicable across multiple research and development contexts:
1. Drug Metabolism and Pharmacokinetics
2. Drug Drug Interaction Evaluation
3. Toxicology and Safety Assessment
4. Mechanistic Studies
5. Biotransformation Research
FAQ
Q1: What types of samples are suitable?
Our UGT Induction Assays support a wide range of small molecule compounds intended for metabolic, pharmacokinetic, or drug drug interaction evaluation. Suitable samples include early discovery leads, clinical candidates, metabolites, reference controls, and investigational chemical entities. Compounds should be soluble in appropriate cell compatible solvents and remain stable during incubation with primary hepatocytes. If your molecule has limited solubility or special handling requirements, our team can assist in selecting the most appropriate formulation strategy.
Q2: How should I prepare my samples?
To ensure reliable induction assessment, samples should be provided in high purity and accompanied by basic physicochemical information. We recommend preparing stock solutions at known concentrations in DMSO or another validated solvent, ensuring that the final solvent percentage in culture remains within acceptable limits. Compounds should be aliquoted to avoid repeated freeze thaw cycles and shipped with clear labeling, safety data, and handling instructions. If the compound is light sensitive, volatile, or unstable, please indicate all relevant storage and protection requirements.
For more information, please refer to Sample Submission Guidelines for Proteomics and Sample Submission Guidelines for Metabolomics.
Q3: What is the service general workflow?
Q4: What data formats are provided?
We supply a complete data package in widely used formats to support downstream analysis and regulatory review:
1. PDF reports summarizing the full study workflow, induction results, data interpretation, and quality control information.
2. Excel or CSV files containing processed and raw numerical data, including qPCR values, fold changes, enzyme activity results, and dose response tables.
3. LC-MS raw and processed output files provided in standard instrument compatible formats for independent verification and further analysis.
4. High resolution image files such as PNG or TIFF for graphs, induction curves, and analytical figures.
5. Additional file formats can be provided upon request to accommodate publication needs or custom data processing workflows.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs delivers reliable, data driven UGT induction assessment supported by high quality hepatocyte models and advanced analytical platforms. Our integrated approach provides clear, actionable insights to guide metabolic risk evaluation and support confident decision making throughout drug development.
How to order?







