Physiologically Based Pharmacokinetic (PBPK) Modeling Service
- Uses mechanistic compartments to represent drug movement among tissues.
- Supports estimation of transfer constants and organ-specific distribution behavior.
- Helps interpret peptide absorption, distribution, and clearance patterns.
- Provides empirical calculations of AUC, clearance, half-life, and volume of distribution.
- Serves as an initial evaluation step for integrating available experimental data into PBPK models.
- Evaluates pharmacokinetic variability across populations.
- Identifies patient-specific covariates that influence drug exposure.
- Supports dose optimization, clinical trial simulation, and patient subgroup prediction.
MtoZ Biolabs provides a Physiologically Based Pharmacokinetic (PBPK) Modeling Service to support data-driven prediction of peptide absorption, distribution, metabolism, and excretion properties using mechanistic computational models. Our platform integrates physiological databases and physicochemical parameters to simulate in vivo behavior of peptide drug. This service offers predictive insights essential for optimizing peptide drug candidates, planning preclinical studies, and guiding translational decisions.
What Is Physiologically Based Pharmacokinetic (PBPK) Model?
Physiologically Based Pharmacokinetic modeling is a computational approach that describes how a compound moves through the body using mathematical representations of tissue compartments, physiological parameters, and biochemical processes. Instead of relying solely on empirical observations, PBPK integrates known biological features such as organ blood flow, tissue composition, metabolic capacity, and transport mechanisms. This structure allows PBPK models to simulate complex pharmacokinetic behavior and predict drug performance under various physiological or pathological conditions.
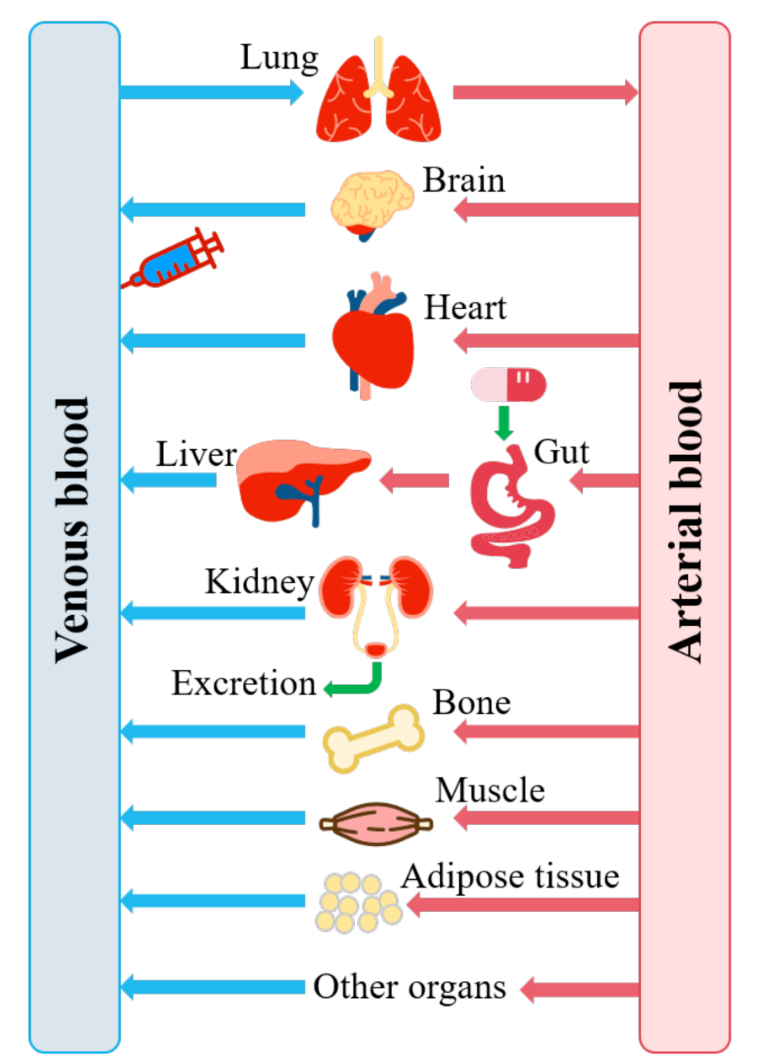
Peng, Y. et al. Metabolites. 2021.
Figure 1. A Generic Physiologically Based Pharmacokinetic (PBPK) Model
PBPK models enable researchers to forecast concentration-time profiles, predict exposure in different tissues, estimate oral bioavailability, and assess how intrinsic or extrinsic variations influence drug behavior. These models can also support decision making related to dosing, formulation design, drug interaction assessment, pediatric or geriatric extrapolation, and regulatory submissions.
Physiologically Based Pharmacokinetic (PBPK) Modeling Service at MtoZ Biolabs
MtoZ Biolabs offers a robust PBPK Modeling Service specifically aligned with the needs of peptide therapeutics. Our solutions combine computational modeling, pharmacokinetic data processing, and predictive analytics to support development activities ranging from lead selection to clinical planning.
Integrated Pharmacokinetic Analysis Approaches
We build PBPK models using a comprehensive combination of analytical frameworks:
🔸Compartmental Analysis
🔸Non-Compartmental Analysis (NCA)
🔸Population Pharmacokinetic Analysis (PopPK)
By combining these complementary approaches, MtoZ Biolabs creates highly reliable PBPK models with strong predictive accuracy.
Key Predictive Services We Provide
Our PBPK Modeling Service includes capabilities essential for peptide drug development:
💠PK and PD Predictions
Simulation of exposure profiles, pharmacodynamic outcomes, and dose-response behavior.
💠ADMET Predictions
Assessment of absorption, metabolism, excretion, tissue deposition, and toxicity risks using integrated mechanistic modeling.
💠Drug-Drug Interaction Predictions
Identification of potential interactions based on enzyme modulation, transporter involvement, or metabolic pathway saturation.
💠Oral Bioavailability Prediction for Peptide Drugs
Evaluation of GI permeability, enzymatic degradation, and formulation effects to determine viability of oral administration strategies.
These services enable clients to make data-driven decisions about formulation design, dosing regimens, and clinical strategies with confidence.
Why Choose MtoZ Biolabs
✔️Extensive Experience
Our team has extensive experience addressing the unique pharmacokinetic challenges inherent to peptides.
✔️Advanced Computational Tools
We employ high-performance modeling platforms and validated PK analysis methods.
✔️High Predictive Accuracy
Our iterative optimization process ensures strong alignment between predicted and observed data.
✔️Flexible Study Design
Models can be customized to support early discovery, preclinical development, or clinical translation.
✔️Dedicated Scientific Support
Experts provide continuous communication, data interpretation, and guidance.
Applications of Physiologically Based Pharmacokinetic (PBPK) Modeling Service
1. Dose Selection and Optimization
Supports calculation of safe and effective dosing regimens for early research or clinical design.
2. Mechanistic Assessment of ADMET
Evaluates peptide absorption limitations, metabolic degradation, clearance pathways, and tissue penetration.
3. Drug Interaction Risk Assessment
Predicts changes in exposure when co-administered with inhibitors, inducers, or competing substrates.
4. Route of Administration Evaluation
Determines feasibility of alternative routes such as oral, subcutaneous, or intranasal delivery.
FAQ
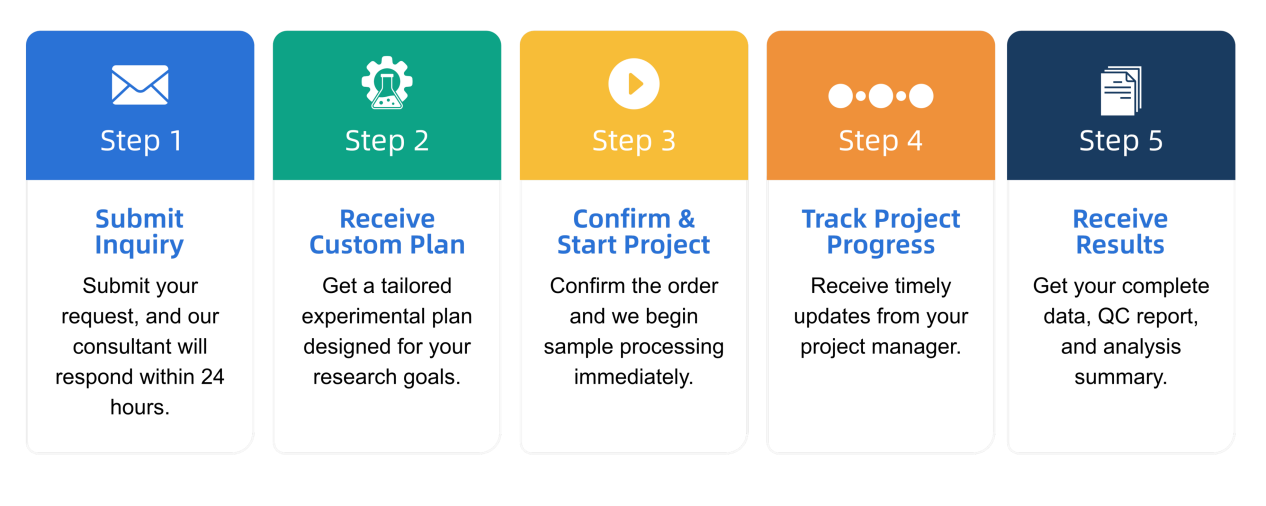
Q1: What is the service general workflow?
Q2: What data formats are provided?
MtoZ Biolabs delivers modeling outputs in standard, analysis-ready formats:
1. PDF reports containing model structure, assumptions, validation, and simulation outcomes
2. Excel or CSV files for parameter tables, PK metrics, and scenario results
3. High-resolution PNG or TIFF figures for concentration-time curves, diagnostic plots, and sensitivity analyses
4. Additional formats can be provided according to project requirements.
Start Your Project with MtoZ Biolabs
By combining compartmental, non-compartmental, and population PK methods, MtoZ Biolabs delivers accurate predictions that strengthen decision making across research and development stages. Our service enhances understanding of peptide pharmacokinetics, supports clinical translation, and empowers drug developers with reliable quantitative tools.
If you would like to initiate a PBPK modeling project or discuss data requirements, please feel free to contact us. MtoZ Biolabs is ready to provide expert consultation and customized support.
How to order?







