Peptide Impurity and Degradation Product Analysis Service
MtoZ Biolabs offers Peptide Impurity and Degradation Product Analysis Service designed to evaluate the structural integrity and purity of peptide materials throughout development and storage. This service supports researchers in identifying impurity species, monitoring degradation behavior, and ensuring the quality attributes required for peptide research, optimization, and product development.
Overview
Peptide impurities and degradation products can arise from incomplete synthesis, sequence-related anomalies, or chemical instabilities such as oxidation, deamidation, hydrolysis, or backbone cleavage. These species may impact biological activity, stability, safety, and consistency, making their assessment essential during formulation design, manufacturing refinement, and long-term storage planning.
Impurity analysis focuses on identifying unintended variants and determining their origins and abundance, while degradation profiling examines how peptides change under real-time, accelerated, or stress conditions. Together, these analyses provide a clear understanding of peptide behavior and help researchers evaluate physicochemical robustness across development stages.
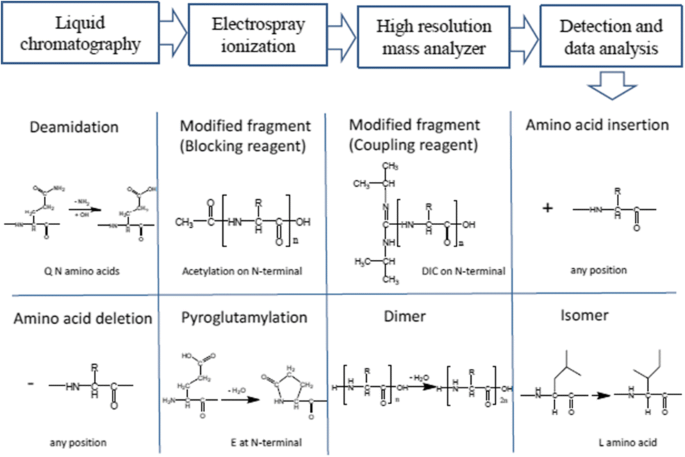
Li, M. et al. Anal Bioanal Chem. 2018.
Figure 1. LC-MS Workflow and Common Structural Variants in Peptide Impurity and Degradation Analysis
Peptide Impurity and Degradation Product Analysis Service at MtoZ Biolabs
Our Peptide Impurity and Degradation Product Analysis Service employs high-resolution LC-MS/MS, stability-indicating chromatographic methods, and fragmentation-based structural elucidation to characterize impurity components and degradation pathways with precision. These analytical platforms allow us to detect, identify, and quantify:
Synthetic byproducts, including deletion sequences, insertion variants, truncated or elongated peptides, and incomplete coupling products
Chemical modification impurities, such as oxidation, deamidation, isomerization, disulfide scrambling, and side-chain alterations
Process-related impurities, including protecting-group remnants, reagent residues, and resin-derived contaminants
Degradation products generated under thermal, pH, oxidative, accelerated, or stress testing conditions
This integrated analytical approach provides detailed impurity fingerprints and degradation route insights that support peptide stability studies, formulation development, and quality control strategies.
Why Choose MtoZ Biolabs?
✅ Broad Impurity Detection Scope
We identify synthetic impurities and degradation products ranging from truncated peptides to oxidation, hydrolysis, and deamidation forms.
✅ High-Resolution Structural Identification
Our advanced LC-MS platform provides precise structural clarification, enabling confident differentiation of closely related peptide variants.
✅ Tailored Workflows
Methods are customized for linear, cyclic, modified, cysteine-rich, and formulated peptides to ensure accurate impurity profiling.
✅ Expert Scientific Support
Our team offers guidance on impurity risks, testing strategies, and formulation considerations throughout the project.
✅ One-Time-Charge
Our pricing is transparent, no hidden fees or additional costs.
Applications of Peptide Impurity and Degradation Product Analysis Service
This service is well-suited for:
· Evaluating peptide purity during research and early development
· Tracking degradation behavior under real-time and accelerated stability conditions
· Supporting formulation and process optimization
· Investigating batch-to-batch consistency and troubleshooting manufacturing issues
· Preparing impurity profiles for quality documentation and regulatory submissions
Start Your Project with MtoZ Biolabs
MtoZ Biolabs provides precise and comprehensive impurity and degradation product profiling to support peptide quality control, formulation development, and regulatory readiness. Contact us to begin your analysis or discuss custom project requirements.
FAQ
Q1: What types of samples are suitable?
We accept synthetic peptides, peptide APIs, formulated peptides, peptide-drug conjugates, and stability or stress-study samples across a range of purity levels. Both linear and cyclic peptides, modified peptides, and formulation-containing materials are compatible with our analytical workflows.
Q2: What is the service general workflow?
Q3: What data formats are provided?
Deliverables include LC-MS/MS chromatograms, impurity and degradation product profiles, mass spectral data, quantitative tables, and a comprehensive report summarizing results and interpretations.
All data are provided in standard formats (Excel, PDF, and instrument raw files) suitable for publication and further bioinformatics analysis.
Q4: How should I prepare my samples?
· Submitting samples in clean, properly labeled vials
· Providing 1-2 mg of peptide material for full impurity and degradation profiling
· Including information on formulation components, known modifications, or storage history
· Shipping samples with appropriate temperature control to maintain integrity
If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







