Peptide Drug In Vitro Cytotoxicity Assessment Service
MtoZ Biolabs offers Peptide Drug In Vitro Cytotoxicity Assessment Service to evaluate the safety and biological compatibility of peptide-based therapeutics. Cytotoxicity testing is a critical step in identifying potential risks such as cell death or toxicity before advancing peptides into preclinical or clinical development. Our service uses advanced cell-based assays to provide reliable, reproducible data that inform peptide selection, optimization, and development.
Overview
Peptide drug in vitro cytotoxicity assessment is essential to evaluate the potential toxic effects of peptide therapeutics on living cells. This testing identifies whether a peptide induces harmful biological responses, such as cell death, apoptosis, or membrane disruption. Cytotoxicity assessments are carried out in vitro using cultured mammalian cells, ensuring relevance to human physiology.
This testing is crucial for determining whether a peptide candidate will cause undesirable effects in humans, ensuring it is safe for further development. It helps identify peptides with low toxicity profiles, guiding the selection of safe candidates and optimal formulations for clinical advancement.
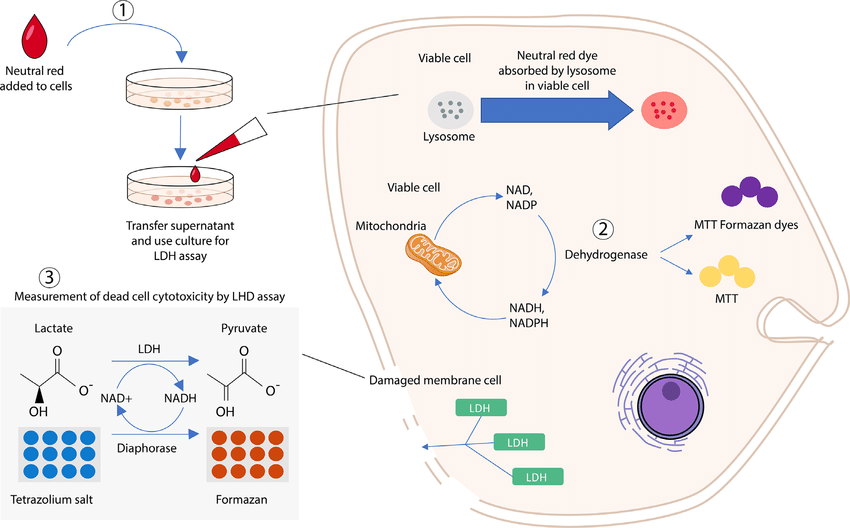
Soltani, S. et al. FEMS Microbiol Rev. 2021.
Figure 1. In Vitro Cytotoxicity Assays
Peptide Drug In Vitro Cytotoxicity Assessment Service at MtoZ Biolabs
MtoZ Biolabs offers a comprehensive suite of Peptide Drug In Vitro Cytotoxicity Assessment services designed to address the full range of cytotoxicity evaluation needs:
1. Cytotoxicity and Dose-Response Evaluation
Quantitative assessment of peptide-induced toxicity, including IC50 determination and concentration-dependent effects.
2. Cell Viability and Growth Analysis
Measurement of cellular metabolic activity, proliferation, and overall health following peptide exposure.
3. Apoptosis and Mechanism-Based Toxicity Profiling
Characterization of cell death pathways through caspase activation, apoptosis detection, and membrane stability assessment.
4. Formulation Compatibility and Cytotoxicity Testing
Evaluation of peptide formulations, excipients, and delivery systems to ensure biological compatibility.
5. High-Throughput Cytotoxicity Screening for Peptide Libraries
Scalable platforms for rapid assessment of peptide variants or large compound sets.
A wide selection of 2D and advanced 3D culture models is available upon request, including epithelial, fibroblast, hepatocyte, and co-culture systems.
Why Choose MtoZ Biolabs?
✅ Comprehensive Assay Portfolio: Multiple validated cytotoxicity assays, suitable for both rapid screening and detailed characterization.
✅ Advanced Analysis Platform: High-content imaging and biochemical readouts enable sensitive detection of peptide-induced cellular responses.
✅ Multimodel Compatibility: Assay options for various 2D and 3D cell systems to support physiologically relevant evaluation.
✅ Experienced Scientific Team: Extensive expertise in peptide analysis, assay design, and data interpretation.
✅ One-Time-Charge: Our pricing is transparent, no hidden fees or additional costs.
Start Your Project with MtoZ Biolabs
Support your peptide development program with accurate and comprehensive cytotoxicity analyses. MtoZ Biolabs provides high-quality Peptide Drug In Vitro Cytotoxicity Assessment Service data to guide peptide optimization, safety evaluation, and preclinical readiness.
Contact us to initiate your cytotoxicity testing program.
FAQ
Q1: What types of samples are suitable?
We accept linear peptides, cyclic peptides, peptide conjugates, peptidomimetics, and formulated peptide products. Both research-grade and preclinical-grade samples are compatible with our assay systems.
Q2: What is the service general workflow?
Q3: What data formats are provided?
We supply raw assay data, processed datasets in Excel or CSV format, dose-response summaries, IC50 results, and comprehensive interpretive reports.
Q4: How should I prepare my samples?
To ensure reliable testing:
Store samples at -80℃
Avoid repeated freeze-thaw cycles
Provide adequate sample volume for all planned assays
Include information on formulation components, buffers, or excipients
Clarify samples by centrifugation when necessary
Ship samples on dry ice using insulated packaging
For more information, please refer to Sample Submission Guidelines for Proteomics. If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







