Peptide Drug Genotoxicity Assessment Service
MtoZ Biolabs provides comprehensive Peptide Drug Genotoxicity Assessment Service to support early evaluation of potential genetic risks associated with peptide therapeutics. Genotoxicity studies help identify whether peptide candidates may induce DNA damage, genetic mutations, or chromosomal alterations, offering important safety insights before advancing into later development stages.
Overview
Peptide Drug Genotoxicity Assessment focuses on identifying whether a peptide or its degradation products interact with genetic material or alter cellular processes responsible for maintaining genomic stability. While peptides are often metabolically labile, certain structural features, reactive groups, or conjugation elements may influence their potential for genotoxic interactions.
Genotoxicity testing typically examines gene mutation frequency, chromosomal integrity, and DNA strand damage using validated in vitro and in vivo systems. These studies generate foundational safety information that supports candidate ranking, risk assessment, and preparation for more detailed toxicology evaluations. Early detection of genotoxic concerns helps streamline development and reduce the likelihood of advancing unsuitable candidates into later stages.
Peptide Drug Genotoxicity Assessment Service at MtoZ Biolabs
MtoZ Biolabs performs a range of genotoxicity studies relevant to peptide drug discovery and early development, including:
· Bacterial Reverse Mutation Test (Ames Test)
Evaluation of gene mutation potential across multiple bacterial strains with and without metabolic activation.
· In Vitro Micronucleus Test
Detection of chromosomal breakage or loss in mammalian cell systems.
· In Vitro Mammalian Chromosome Aberration Test
Identification of structural chromosomal abnormalities under controlled exposure conditions.
· In Vitro Mammalian Cell Gene Mutation Test
Analysis of forward or reverse mutations at defined gene loci in standard mammalian cell lines.
· In Vivo Micronucleus Test
Assessment of chromosomal damage in bone marrow or peripheral blood following peptide administration.
· Comet Assay (In Vitro or In Vivo)
Measurement of DNA strand breaks and repair-associated responses in mammalian cells or tissues.
Why Choose MtoZ Biolabs?
✅ Practical experience supporting a broad range of in vitro and in vivo assays relevant to the early safety evaluation of peptide therapeutics.
✅ Validated bacterial strains, mammalian cell lines, and DNA damage assays that enable consistent assessment of genetic integrity.
✅ Organized operational workflows that support reliable sample handling, exposure procedures, and endpoint evaluation.
✅ Results delivered in a structured format that supports comparison across dose groups and assists early development decision-making.
✅ Predictable scheduling, steady communication, and dependable support throughout the duration of each study.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs offers structured and reliable genotoxicity assessments designed to support early-stage peptide drug development. Our team is ready to assist with planning and execution of genetic safety studies tailored to your program’s needs.
Contact us to begin your Peptide Drug Genotoxicity Assessment Service.
FAQ
Q1: What types of samples are suitable?
Peptides composed of natural amino acids, non-natural amino acids, linker-containing structures, peptide conjugates, peptidomimetics, and other peptide-derived chemical entities may be evaluated when purity, identity, and stability data are available.
Q2: What is the service general workflow?
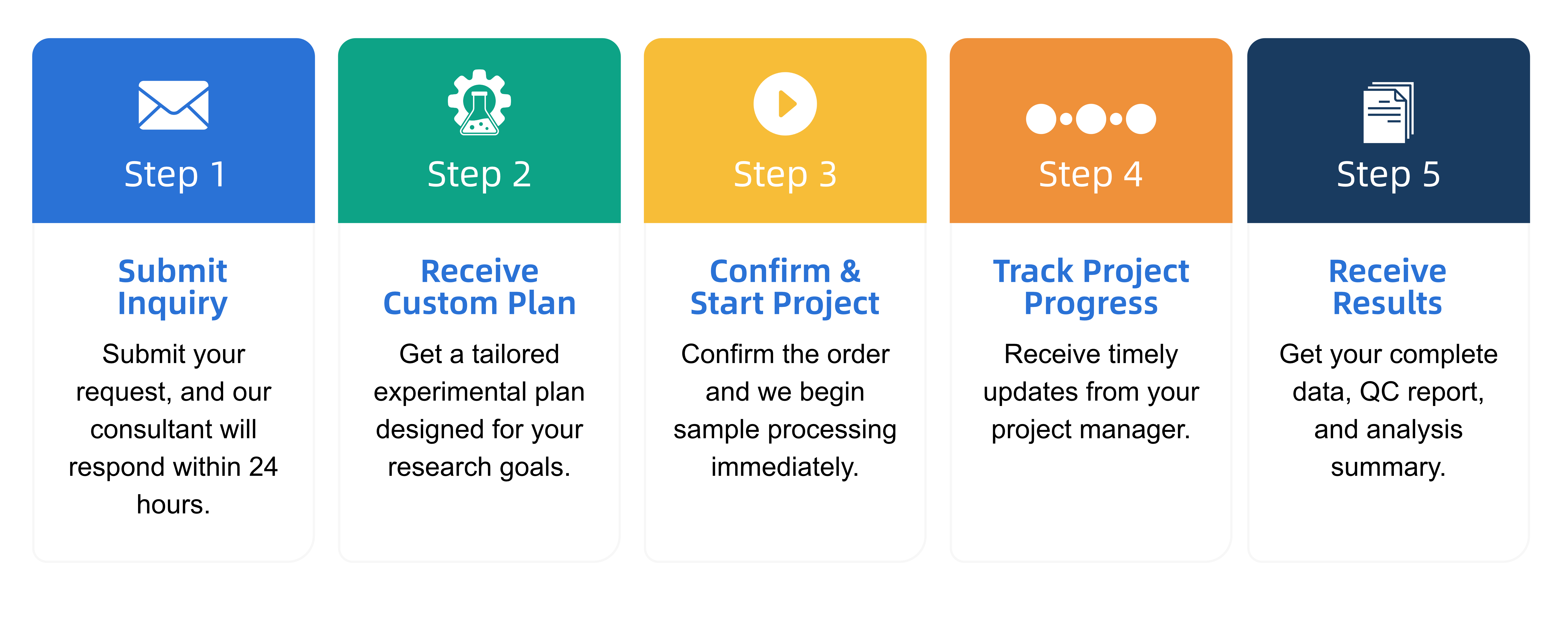
Q3: What data formats are provided?
Deliverables include raw data files, mutation frequency results, chromosomal assessment summaries, comet assay outputs, and full study reports in PDF, Excel, and CSV formats.
Q4: How should I prepare my samples?
· Provide test materials with confirmed purity and identity
· Include formulation components, solubility information, and storage recommendations
· Supply enough material to support all planned test concentrations
· Ship samples under appropriate temperature-controlled conditions
· Inform the study team of any special handling or stability considerations
For more information, please refer to Sample Submission Guidelines for Proteomics. If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







