Peptide Drug Cardiotoxicity Assessment Service
MtoZ Biolabs provides Peptide Drug Cardiotoxicity Assessment Service to support the early evaluation of cardiac safety risks associated with peptide therapeutics. By combining cell-based cardiac models and functional readouts, this service generates reliable data to guide candidate selection, reduce late-stage failures, and enhance safety confidence during drug development.
Overview
Cardiotoxicity is a major contributor to compound attrition and clinical withdrawal. Peptide drugs, while often designed for selective mechanisms, may influence key cardiac pathways involving electrophysiology, contractility, or cellular metabolism. Early detection of these risks is essential to avoid unexpected cardiac effects.
Cardiac safety profiling helps assess:
· Changes in cardiomyocyte viability, morphology, or structural integrity
· Electrophysiological disturbances or altered membrane behavior
· Disrupted calcium handling and ion flux
· Mitochondrial stress and metabolic impairment
· Early indicators of hypertrophy or contractile dysfunction
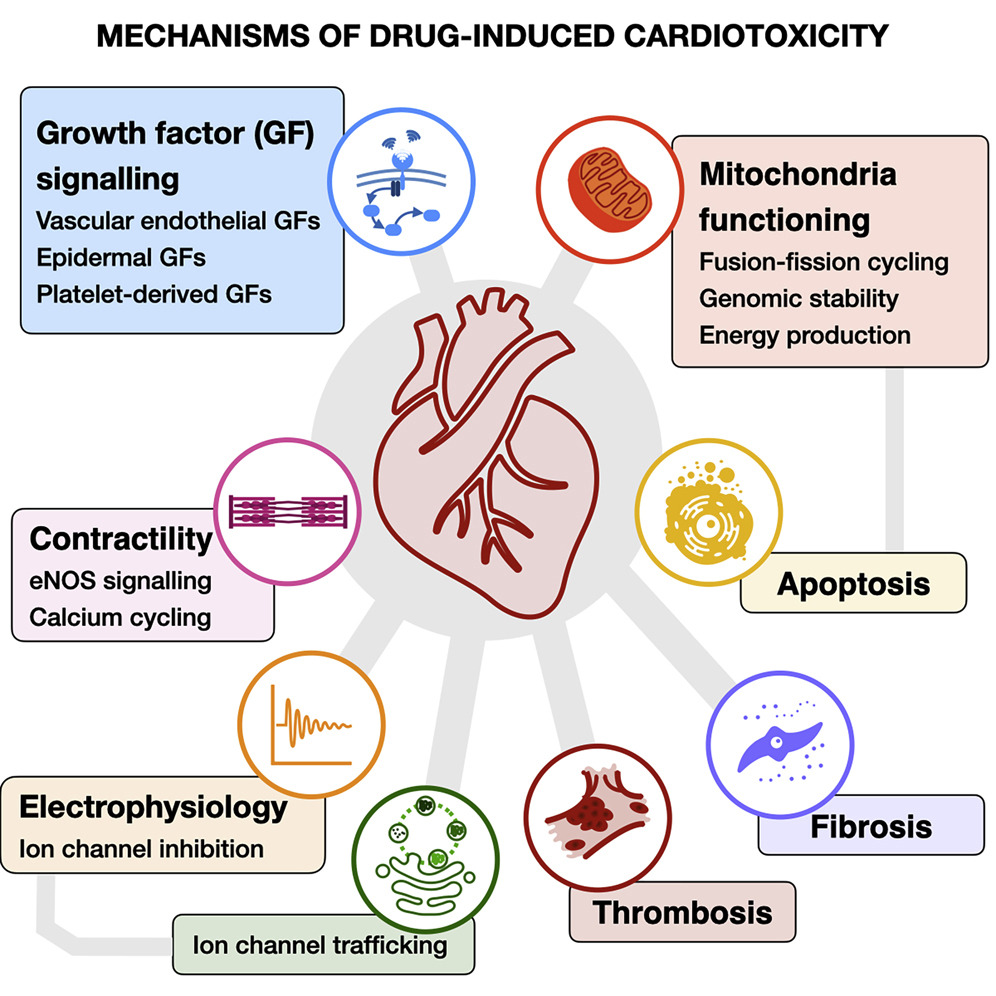
Mamoshina, P. et al. Cell Rep Med. 2021.
Figure 1. Mechanisms of Drug-induced Cardiotoxicity
Peptide Drug Cardiotoxicity Assessment Service at MtoZ Biolabs
MtoZ Biolabs offers a focused and industry-aligned Peptide Drug Cardiotoxicity Assessment Service that includes commonly used approaches suitable for peptide safety evaluation. Our service options include:
1. Cardiomyocyte viability and morphology analysis
Assessment of structural changes, cell health, and cytotoxicity using established cardiac cell systems.
2. Electrophysiological and functional evaluation
Measurement of electrical and contractile properties to detect early functional disturbances.
3. Calcium flux and ion handling assays
Evaluation of calcium dynamics to identify changes in cardiomyocyte signaling and contraction.
4. Mitochondrial function and metabolic stress testing
Characterization of mitochondrial performance and metabolic responses to peptide exposure.
5. Cardiac histopathology
Examination of cardiac tissues in validated animal models to evaluate structural or cellular alterations.
6. Biomarker-based cardiac injury assessment
Measurement of relevant cardiac injury markers for supportive safety insights.
Why Choose MtoZ Biolabs?
● Rapid in vitro screening formats, including fluorescence-based hERG assessments and contractility pre-screens, support efficient evaluation of multiple peptide variants.
● Extensive experience studying cardiac responses to linear peptides, cyclic peptides, conjugated peptides, and receptor-targeted peptide constructs.
● Structured interpretation that consolidates electrophysiology findings, cellular responses, biomarker measurements, and tissue observations into clear, decision-oriented summaries.
● Organized scheduling, consistent communication, and predictable study management ensure smooth progression from sample receipt to report delivery.
● Our pricing is transparent, no hidden fees or additional costs.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs is committed to delivering high-quality Peptide Drug Cardiotoxicity Assessment Service to accelerate peptide drug development and support confident safety decisions. Contact us to begin planning your study.
FAQ
Q1: What types of samples are suitable?
Suitable sample types include lyophilized peptides, peptide solutions, cyclic peptides, peptide–drug conjugates, receptor-targeted peptides, and other modified peptide constructs. Typical needs are 1-2 mg peptide or 1-2 mL of sample. For unconventional formats, our team can evaluate feasibility before submission.
Q2: What is the service general workflow?
Q3: What data formats are provided?
Data are delivered in standard, analysis-ready formats including PDF study reports, Excel or CSV data tables, raw electrophysiology files, imaging files for structural assessments, and export formats compatible with common analytical software.
Q4: How should I prepare my samples?
· Prepare samples at concentrations appropriate for planned assays.
· Use high-purity materials without interfering excipients.
· Clearly label each vial with concentration, formulation, and batch information.
· Freeze samples promptly and minimize freeze–thaw cycles.
· Ship on dry ice using insulated packaging to maintain stability.
For more information, please refer to Sample Submission Guidelines for Proteomics. If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







