Peptide Drug Carcinogenicity Assessment Service
MtoZ Biolabs provides Peptide Drug Carcinogenicity Assessment Service to evaluate whether peptide candidates have the potential to induce or promote tumor formation during development. Carcinogenicity evaluation is an essential component of peptide drug safety assessment, supporting risk identification, candidate prioritization, and early decision-making. By integrating validated in vitro and in vivo approaches, MtoZ Biolabs delivers reliable and well-interpreted data that help clients advance peptide drug programs with confidence.
Overview
Carcinogenicity assessment focuses on determining whether a compound can initiate or influence neoplastic processes. Although peptides generally display favorable safety profiles, certain peptide classes, including hormone analogs or receptor agonists, may influence cell proliferation pathways. As a result, screening for carcinogenicity is required to ensure that long-term administration does not elevate tumor risk.
In vitro assays offer insights into mutagenicity and proliferative potential, while in vivo studies allow observation of systemic and tissue-specific outcomes. Together, these approaches provide a comprehensive understanding of carcinogenic risks and support peptide drug development across discovery and nonclinical stages.
Peptide Drug Carcinogenicity Assessment Service at MtoZ Biolabs
MtoZ Biolabs offers a structured portfolio of carcinogenicity assessment services, integrating both in vitro and in vivo approaches to support a wide range of peptide drug modalities.
1. In vitro carcinogenicity assessment
Includes mutagenicity screening, peptide-induced proliferative activity evaluation, and chromosomal damage assays to detect early carcinogenic signals at the cellular level.
2. In vivo carcinogenicity assessment
Covers rodent-based evaluations of systemic genotoxicity, proliferative responses, and tumorigenic potential using established toxicology models.
Why Choose MtoZ Biolabs?
1. Integrated carcinogenicity assessment: Combined in vitro and in vivo strategies enable comprehensive evaluation of tumor-related risks.
2. Advanced scientific expertise: Experienced toxicologists support study planning, data interpretation, and peptide-specific risk analysis.
3. High-quality analytical standards: All studies follow stringent scientific criteria for reliability, reproducibility, and traceability.
4. Flexible study design: Customizable workflows accommodate different peptide classes, mechanisms of action, and development objectives.
5. Responsive project management: Efficient communication and coordinated planning support predictable timelines and streamlined execution.
Start Your Project with MtoZ Biolabs
Advance your safety evaluation with MtoZ Biolabs. Our Peptide Drug Carcinogenicity Assessment Service offers the focused support needed to move candidates confidently into nonclinical stages. Contact us to outline your study requirements and initiate your project.
FAQ
Q1: What types of samples are suitable?
MtoZ Biolabs accepts synthetic peptides, peptide analogs, and formulation-ready peptide samples. A typical submission includes milligram-scale quantities depending on the scope of the study.
Q2: What is the service general workflow?
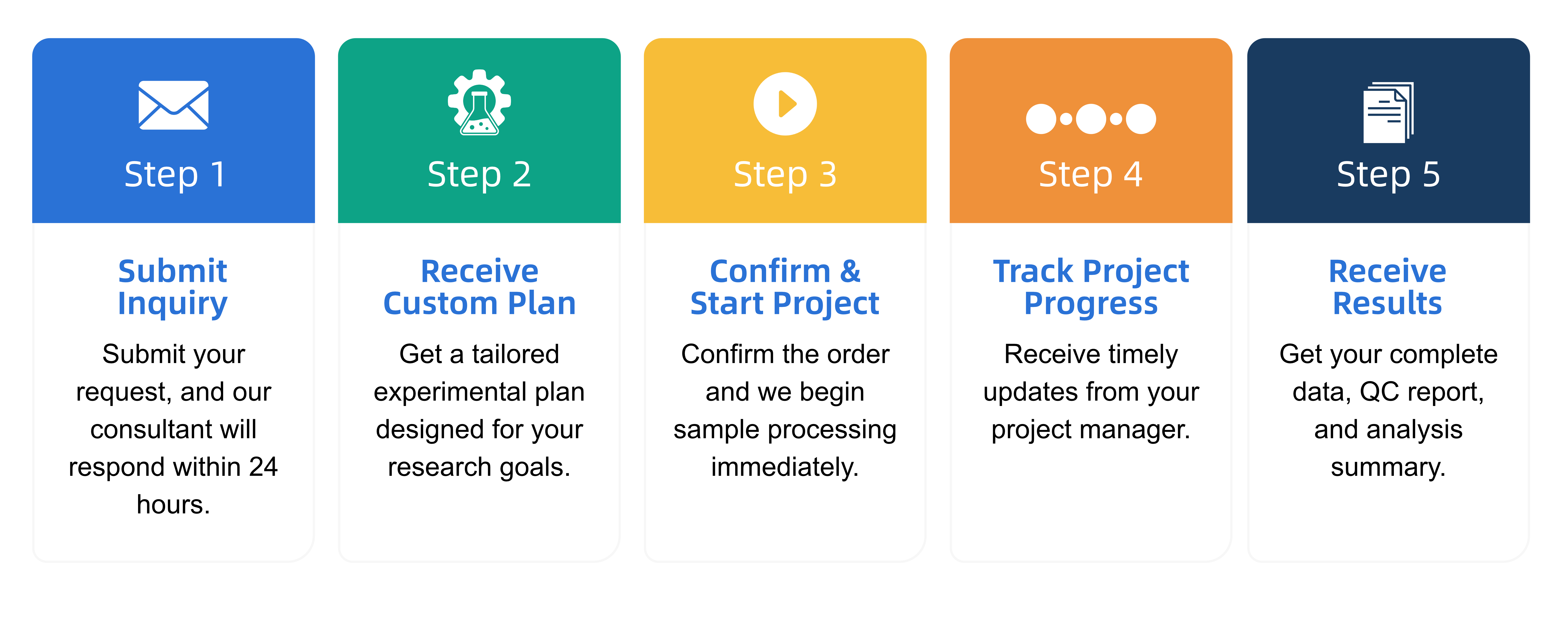
Q3: What data formats are provided?
Raw instrument outputs from in vitro assays and in vivo observations
Processed data tables in Excel or CSV formats
Dose-response curves, proliferation profiles, and statistical analyses
Histopathology findings and imaging data (TIFF/PNG, if applicable)
Comprehensive PDF study reports summarizing methods, results, and carcinogenicity risk interpretation
Custom formatting or additional documentation can be provided upon request to fit your internal review or regulatory submission needs.
Q4: How should I prepare my samples?
Store samples under recommended temperature conditions (e.g., −20°C or −80°C depending on stability)
Avoid repeated freeze-thaw cycles
Provide detailed sample information, including concentration, formulation, and storage history
Use appropriate containment (sealed cryovials for peptides, leak-proof tubes for biological matrices)
Ship samples with temperature control (cold packs or dry ice depending on sample type)
If needed, MtoZ Biolabs can provide tailored sample preparation and shipping guidance based on the specific study design.
Related Services
How to order?







