CYP Inhibition Assessment Service
MtoZ Biolabs provides comprehensive CYP Inhibition Assessment Service designed to support drug discovery, early safety evaluation, and pharmacokinetic optimization. Our service focuses on identifying and characterizing the inhibitory effects of drug candidates on major human cytochrome P450 enzymes, enabling researchers to predict metabolic risks and drug drug interaction liabilities with confidence. This service is built for pharmaceutical companies, biotechnology organizations, and academic laboratories seeking reliable and data driven CYP inhibition assessments.
What Is CYP?
Cytochrome P450 enzymes (CYP) constitute a superfamily of heme containing monooxygenases responsible for metabolizing more than seventy percent of clinically used small molecule drugs. Among them, CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A4 are considered the most critical isoforms in human drug metabolism. Inhibition of these enzymes can be reversible, time dependent, competitive, noncompetitive, or mechanism based. Each inhibition type carries different implications for dosing, toxicity, and drug drug interactions.
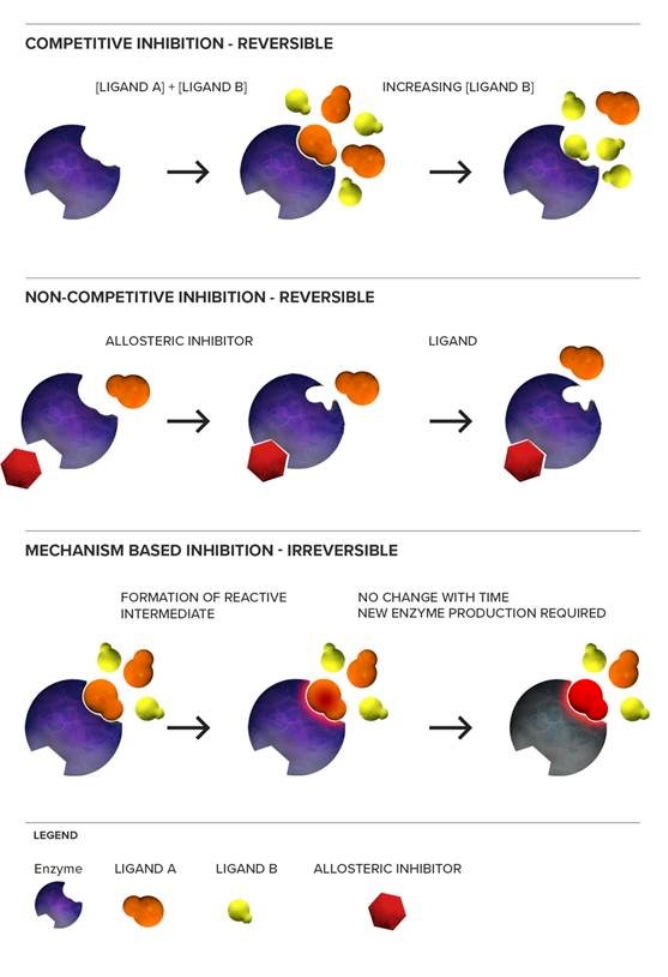
Deodhar, M. et al. Pharmaceutics. 2020.
Figure 1. Mechanisms of CYP450 Inhibition
Understanding a drug candidate's inhibitory potential is essential because metabolic inhibition is one of the primary causes of clinically significant drug interactions. For example, inhibition of CYP3A4 may affect the exposure of drugs with narrow therapeutic windows such as immunosuppressants. Similarly, inhibition of CYP2D6 can impair the biotransformation of antidepressants, antipsychotics, and opioid analgesics. Accurate assessment of CYP inhibition is therefore a fundamental requirement for developing safe and effective therapeutics.
CYP Inhibition Assessment Service at MtoZ Biolabs
Our CYP Inhibition Assessment Service evaluates inhibition against all major human CYP isoforms including CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A4. Assays can be customized to evaluate single isoforms or full CYP panels depending on project requirements. We provide both reversible and time dependent inhibition studies using human liver microsomes, recombinant enzymes, hepatocytes, or client specified systems.
We offer a broad range of CYP inhibition related services including:
💠Reversible Inhibition Assessment
Evaluation of competitive, noncompetitive, mixed mode, and uncompetitive inhibition mechanisms using validated probe substrates.
💠Time Dependent Inhibition Evaluation
Quantification of enzyme inactivation rates, determination of kinact and KI values, and confirmation of mechanism based inhibition.
💠CYP Panel Screening
High throughput or focused panel evaluation across major human CYP isoforms to rapidly classify inhibitory profiles.
💠Enzyme Kinetics Characterization
Calculation of IC50, Ki, and kinetic constants to support medicinal chemistry optimization.
💠Reaction Phenotyping and Inhibition Interaction Studies
Determination of metabolic pathways and identification of which enzymes are most affected by candidate molecules.
💠LC-MS-Based Metabolite Quantification
High resolution LC-MS workflows for accurate detection of probe substrate turnover and metabolite formation.
Why Choose MtoZ Biolabs
✔️Extensive expertise
Our scientists have deep experience in enzymology, drug metabolism, and LC-MS based quantification.
✔️Advanced analytical instrumentation
High resolution LC-MS platforms and validated probe substrates ensure accurate measurement.
✔️Customizable assay designs
Flexible experimental formats accommodate a wide range of compound classes and study objectives.
✔️Rapid turnaround times
Efficient workflows support fast decision making in early drug discovery.
✔️Integrated interpretation and modeling support
We provide both experimental data and in depth scientific guidance.
Applications of CYP Inhibition Assessment Service
CYP inhibition assessment is crucial across multiple domains of drug development. Key applications include:
1. Drug candidate prioritization
Identify compounds with minimal metabolic liability early in discovery.
2. Drug drug interaction risk evaluation
Predict clinical interactions with coadministered therapeutics.
3. Medicinal chemistry optimization
Guide structural modifications to reduce inhibitory potency.
4. Pharmacokinetic modeling
Provide kinetic parameters for PBPK simulation and exposure prediction.
5. Safety assessment programs
Assess toxicity potential arising from inhibited metabolic pathways.
FAQ
Q1: What types of samples are suitable?
The service is suitable for purified small molecule drug candidates, peptide based compounds, and reference standards intended for CYP inhibition analysis. We also accept compound libraries for panel screening projects. Samples should be provided in a stable, well characterized form.
Q2: How should I prepare my samples?
Samples should be supplied as purified solids or solutions with clearly indicated concentration, solvent composition, and stability requirements. Compounds must be free of particulate matter and compatible with microsomal or recombinant enzyme assays. Tissue or crude extracts are not recommended for CYP inhibition studies.
For more information, please refer to Sample Submission Guidelines for Proteomics and Sample Submission Guidelines for Metabolomics
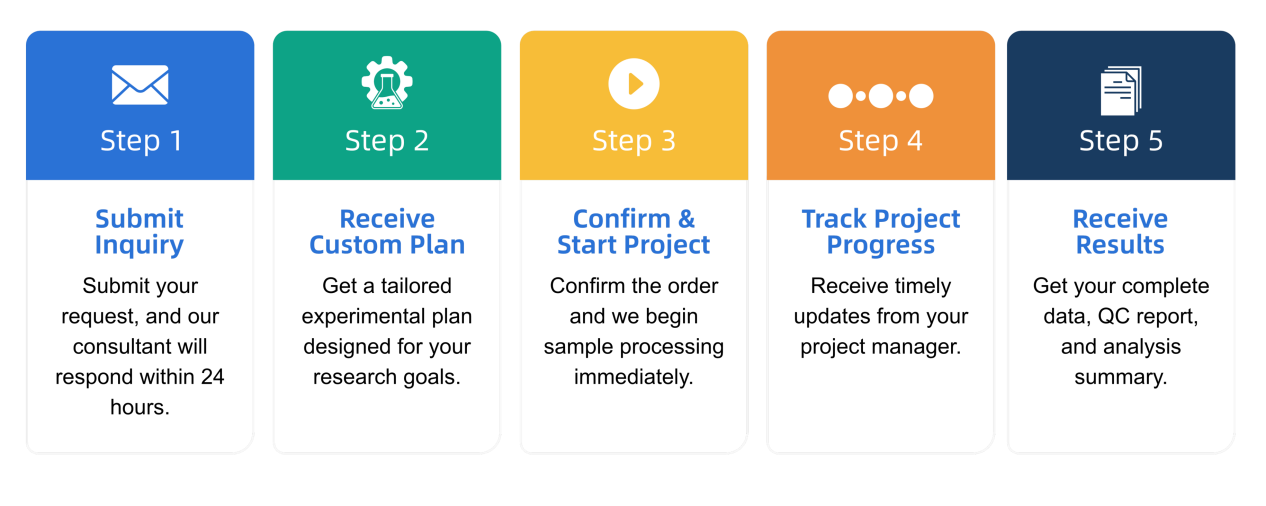
Q3: What is the service general workflow?
Q4: What data formats are provided?
We deliver a consolidated PDF report detailing assay parameters and inhibition outcomes, complemented by Excel or CSV files containing processed kinetic data and calculated IC values. Raw chromatographic or mass spectrometric outputs can be provided upon request in standard instrument compatible formats to support further analysis.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs offers a complete suite of CYP inhibition assessment solutions that blend state of the art instrumentation with standardized regulatory compliant workflows. Our goal is to help clients identify potential metabolic liabilities early and support decision making across medicinal chemistry, pharmacokinetics, and safety assessment.
If you are interested in our CYP Inhibition Assessment Service, please feel free to Contact us.
How to order?







