CYP Induction Assessment Service
MtoZ Biolabs provides a comprehensive CYP Induction Assessment Service designed to support drug discovery, preclinical development, and safety evaluation. The CYP Induction Assessment Service focuses on detecting whether drug candidates or research compounds induce cytochrome P450 enzymes, which is a critical factor influencing metabolic stability, exposure, toxicity, and the risk of drug drug interactions. This service uses validated in vitro induction models and high-precision analytical platforms to quantify the impact of compounds on key CYP isoforms and generate mechanistic insights for regulatory decision making.
Background
Cytochrome P450 (CYP) induction is a major regulatory concern in drug development because many xenobiotics can increase the expression or activity of key metabolic enzymes such as CYP1A2, CYP2B6, and CYP3A4. These enzymes play essential roles in hepatic metabolism, and their induction may significantly reduce drug exposure, compromise therapeutic efficacy, or lead to interactions with co administered drugs. Understanding CYP induction potential early allows researchers to select safer compounds, and optimize dosing strategies.
CYP Induction Assessment Service offers an in vitro test platform that evaluates whether a compound increases the mRNA, protein, or catalytic activity of P450 enzymes in human-derived systems. Compared with in vivo induction studies, cell-based induction assessments offer faster turnaround time, lower material requirements, ethical feasibility, and improved mechanistic resolution. These methods allow researchers to differentiate between direct metabolic activation and transcriptional regulation events and to identify induction through nuclear receptors such as PXR, CAR, or AhR.
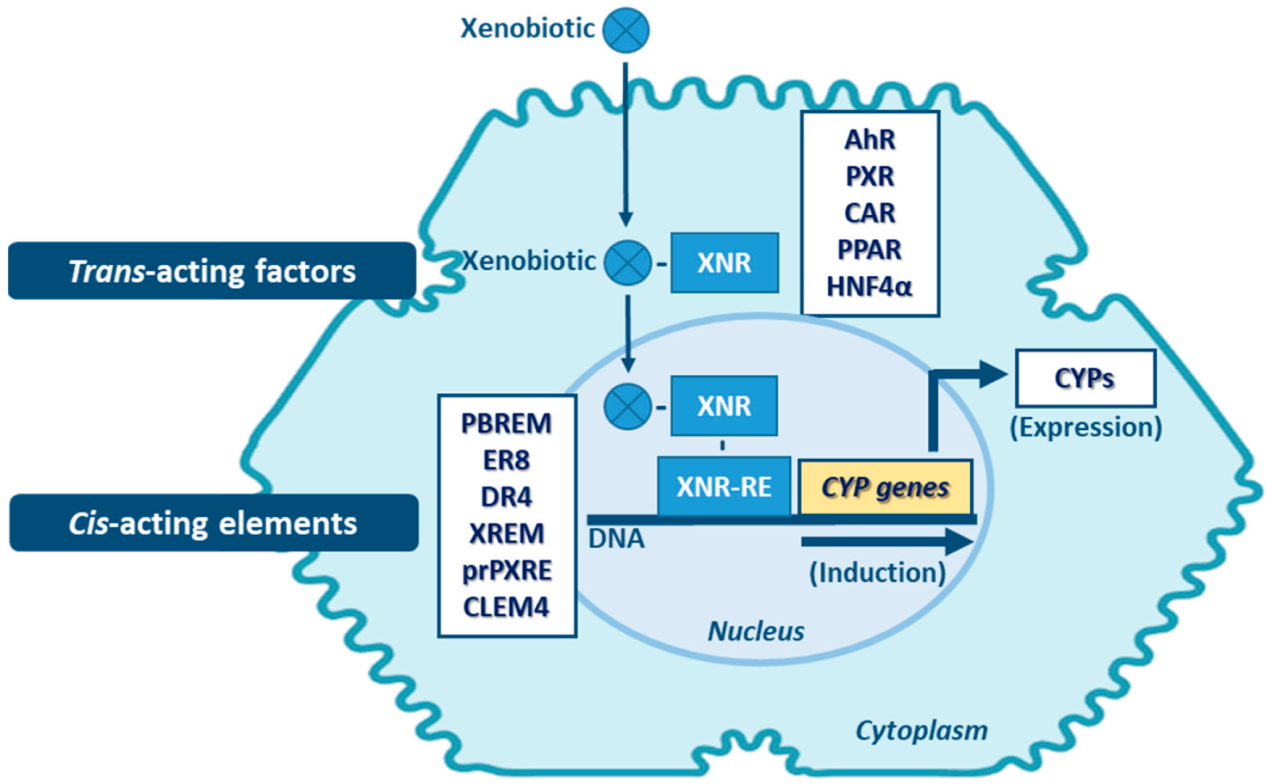
Esteves, F. et al. J Xenobiot. 2021.
Figure 1. Main Mechanisms of CYP Induction
CYP Induction Assessment Service at MtoZ Biolabs
MtoZ Biolabs offers an integrated analytical approach to characterize CYP induction with high accuracy. Our platform combines primary human hepatocytes, nuclear receptor responsive models, and quantitative assays for mRNA, protein, and enzyme activity measurements. The resulting data package supports mechanism-based analysis, decision making, and submission-ready documentation.
Our service helps clients
💠Detect CYP induction reliably
💠Characterize nuclear receptor mediated pathways
💠Assess DDI risks following regulatory guidelines
💠Support medicinal chemistry optimization and compound ranking
Our solutions include
🔸Single isoform induction analysis
🔸Multi-isoform induction profiling
🔸Concentration dependent induction curves
🔸Mechanistic pathway evaluation
🔸Integrated induction and inhibition screening
🔸Full data reporting for regulatory use
Why Choose MtoZ Biolabs
✔️Extensive experience in evaluating CYP modulation using validated methods
✔️Advanced UPLC-MS/MS platforms for highly sensitive metabolic analysis
✔️Flexible study designs tailored to research needs, compound properties, and project stage
✔️High quality data interpretation that links induction outcomes with metabolic implications
✔️Clear reporting formats and efficient turnaround time to support rapid decision making
Applications of CYP Induction Assessment Service
CYP induction assessment is widely used across drug discovery and early development to evaluate metabolic risks and optimize compound selection. Typical applications include:
1. Screening compounds to identify those that may alter the expression or activity of key metabolic enzymes
2. Supporting lead optimization by clarifying metabolic stability and interaction liabilities
3. Characterizing induction profiles to guide dose selection and formulation strategies
4. Evaluating potential drug drug interactions during preclinical studies
5. Comparing metabolic responses across different compound classes or structural series
FAQ
Q1: What types of samples are suitable?
We accept a wide range of biological samples, including but not limited to cell lysates, tissue homogenates, serum, plasma, compound solutions, drug formulations, and purified peptides. Samples can be derived from human, animal, or model organisms. For special materials, please contact MtoZ Biolabs for specific preparation guidance.
Q2: How should I prepare my samples?
1. Provide compounds dissolved in DMSO, aqueous buffer, or another compatible solvent.
2. Ensure samples are clearly labeled and free of particulate matter.
3. Protect light-sensitive or unstable compounds as needed.
For more information, please refer to Sample Submission Guidelines for Proteomics and Sample Submission Guidelines for Metabolomics.
Q3: What is the service general workflow?
Q4: What data formats are provided?
We typically provide activity results, fold-change calculations, and induction curves in Excel or CSV format, along with complete analytical reports in PDF. Raw LC-MS data can be supplied, and additional formats can be provided based on project requirements.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs delivers a scientifically rigorous and regulatory-compliant CYP Induction Assessment Service that enables researchers to evaluate metabolic induction risk with confidence.
Contact us to discuss your project or request a custom quotation.
How to order?







