Which Sample Preparation Methods Improve Histone Phosphorylation Detection?
- Include Na₃VO₄ (sodium vanadate), NaF, and β-glycerophosphate in the lysis buffer
- Perform all steps at low temperature (4°C)
- Minimize the lysis time
- Significantly reduces sample complexity
- Increases the relative proportion of phosphorylated histone peptides in the total sample
- Reduces the burden of subsequent enrichment
- Generates peptides of moderate length that are more suitable for mass spectrometric detection
- Improves the accuracy of phosphorylation site localization
- Reduces interference from coexisting complex modifications
- HCD is suitable for routine phosphorylation analysis.
- ETD is advantageous for multiply phosphorylated peptides.
- DIA improves reproducibility.
In epigenetics and cell signaling research, histone phosphorylation remains challenging to analyze because of its low abundance, highly dynamic nature, and site diversity. Compared with conventional proteomics workflows, the detection of histone phosphorylation is more strongly dependent on sample preparation quality. A poorly designed sample preparation workflow often results in signal loss, limited site coverage, or poor data reproducibility. Which sample preparation methods, then, can significantly improve the efficiency of histone phosphorylation detection?
Rapid, Gentle Cell Lysis Coupled with Phosphatase Inhibition
A key objective during extraction is to prevent the loss of phosphorylation sites. Phosphorylation is highly susceptible to endogenous phosphatase activity. If phosphatase inhibitors are not added promptly during cell lysis, irreversible dephosphorylation may occur, markedly compromising the sensitivity of histone phosphorylation detection.
Optimization recommendations:
This step is critical for subsequent detection sensitivity.
Improving Histone Purity by Acid Extraction
1. Principle
Histones are highly basic proteins and are readily soluble under acidic conditions. Acid extraction using 0.2 M HCl or 0.4 N H₂SO₄ is commonly employed to effectively remove most non-histone proteins from the sample background.
2. Advantages
Compared with direct digestion of whole-cell or whole-protein lysates, acid extraction can markedly improve the detection coverage of phosphorylated histones.
Chemical Derivatization Strategies to Optimize Digestion Efficiency and Improve Histone Phosphorylation Site Resolution
Histones are rich in lysine (Lys) and arginine (Arg). As a result, direct digestion with trypsin often generates excessively short peptides, which are suboptimal for LC-MS/MS analysis.
1. Propionic Anhydride Derivatization (Propionylation)
(1) Principle: Lysine residues are chemically derivatized to prevent excessive tryptic cleavage.
(2) Advantages:
In histone PTM research, derivatization has become a standard component of many analytical workflows.
Optimizing Protease Combinations to Increase Coverage
Different proteases produce distinct cleavage patterns:
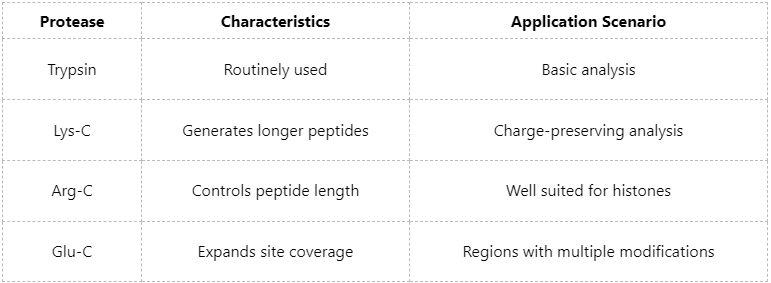
Combined protease digestion strategies can significantly improve phosphorylation site coverage, particularly in regions containing complex modifications.
Desalting and Fractionation Before Phosphopeptide Enrichment
1. StageTip Desalting
This step removes salts and buffer components, thereby improving electrospray ionization efficiency.
2. High-pH Reversed-Phase Fractionation
Fractionation reduces sample complexity and makes low-abundance histone phosphopeptides more likely to be detected. When followed by IMAC or TiO₂ enrichment, it can significantly improve analytical depth.
Selecting Appropriate Phosphorylation Enrichment Strategies
1. IMAC (Immobilized Metal Ion Affinity Chromatography)
Suitable for global analysis of histone phosphorylation and for batch studies involving multiple samples.
2. TiO₂ Enrichment
Suitable for samples with limited starting material and for rapid experimental workflows.
3. Antibody-Based Enrichment
Suitable for mechanistic studies of specific phosphorylation sites and for validation of well-characterized sites such as γ-H2AX. Optimization of the enrichment step directly determines the final sensitivity of phosphorylation detection.
Coordinated Optimization of LC-MS/MS Parameters and Sample Preparation
Sample preparation should be aligned with the mass spectrometry acquisition strategy:
A high-resolution Orbitrap platform combined with an optimized sample preparation workflow can significantly increase both the number and confidence of identified phosphorylation sites.
Common Factors That Affect Detection Efficiency
1. Insufficient Sample Input
For histone phosphorylation analysis, a starting amount of ≥50–100 µg is generally recommended.
2. Excessively Long Processing Time
Phosphorylation is prone to loss during sample handling, and the total processing time should therefore be minimized.
3. Unoptimized Enrichment Conditions
pH and elution buffer composition directly affect enrichment selectivity.
Which sample preparation methods can improve the efficiency of histone phosphorylation detection? The answer does not lie in any single technique, but rather in the systematic optimization of rapid lysis with phosphatase inhibition, acid extraction to improve histone purity, derivatization to optimize proteolysis, rational selection of protease combinations, and coordinated fractionation and enrichment strategies. With the support of a high-quality LC-MS/MS platform, a scientifically designed and well-executed sample preparation workflow can substantially enhance the analytical performance of histone phosphorylation detection. If you are planning a related research project, it is advisable to establish a complete technical workflow at the experimental design stage. Technical support from a professional platform such as MtoZ Biolabs can further improve research depth and the efficiency of downstream translation.
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
How to order?







