Which Factors Affect the Success Rate of Solid-Phase Peptide Synthesis?
-
Hydrophobic amino acids (e.g., Val, Ile, Leu) can promote peptide chain aggregation on the resin, thereby reducing reaction efficiency.
-
Charged amino acids (e.g., Asp, Glu, Arg, Lys) are susceptible to side-chain-related side reactions.
-
Oxidation-prone residues such as Cys, Met, and Trp require specialized protection strategies.
-
Consecutive Pro residues or β-branched amino acids may impose conformational constraints that impede chain elongation.
-
Steric shielding of reactive functional groups
-
Limited accessibility of coupling reagents
-
Incomplete coupling reactions
-
Using low-loading resins
-
Introducing pseudoproline dipeptides
-
Applying the Dmb protecting group strategy
-
Adding aggregation-disrupting additives such as LiCl or 6 M guanidine hydrochloride during coupling
-
HBTU/HOBt
-
HATU
-
DIC/Oxyma
-
PyBOP
-
Aspartimide formation
-
Tryptophan degradation
-
Diketopiperazine formation
-
Conventional synthesis is typically performed at room temperature (20-25°C).
-
Microwave-assisted synthesis (50-90°C) can significantly accelerate reaction rates.
-
However, elevated temperatures may also increase the likelihood of side reactions.
-
TFA 95%
-
TIS 2.5%
-
H₂O 2.5%
-
Low main peak intensity
-
Multiple structurally related impurity peaks
-
Increased difficulty in chromatographic separation
-
LC–MS
-
MALDI-TOF MS
-
High-resolution Orbitrap mass spectrometry
-
Analytical HPLC
-
Molecular weight deviations
-
Missing amino acid residues
-
Oxidative modifications
-
Deletion sequences
-
Perform sequence difficulty prediction prior to synthesis.
-
Optimize resin loading capacity.
-
Adopt double coupling or extended coupling times.
-
Introduce structure-disrupting elements in difficult regions.
-
Strictly control deprotection conditions.
-
Apply high-resolution mass spectrometry for real-time monitoring.
Solid-phase peptide synthesis (SPPS) is a key methodology in modern peptide production and is widely applied in antibody epitope mapping, enzymatic mechanism studies, vaccine development, drug discovery, and protein function analysis. However, in practical laboratory settings, researchers frequently observe that peptide sequences that appear theoretically feasible may encounter various challenges during synthesis, including low coupling efficiency, significant impurity formation, poor product purity, or even complete synthesis failure.
Peptide Sequence Characteristics: The Primary Determinant of Synthetic Difficulty
1. Amino Acid Composition and Sequence Complexity
The physicochemical properties of amino acid residues directly influence coupling efficiency and the likelihood of side reactions during synthesis:
In particular, long peptides (>30 amino acids) or sequences containing repetitive motifs are often classified as difficult sequences, which significantly complicate synthesis.
2. Secondary Structure Propensity and Aggregation Effects
Certain peptide sequences tend to form β-sheet or α-helix-like structures in the solid-phase environment, which can result in:
Common strategies to mitigate these issues include:
Resin Selection: A Critical Role of the Solid Support
Resin selection is a fundamental determinant of SPPS performance, as different resins can significantly influence both product purity and overall yield.
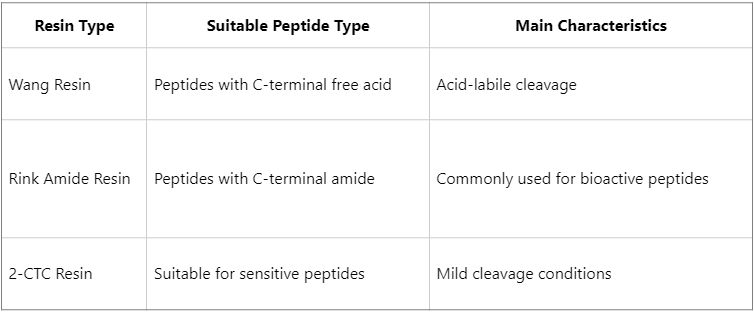
Resin loading capacity is also a crucial parameter. Excessively high loading may lead to steric crowding and peptide aggregation, whereas excessively low loading reduces overall yield. In most cases, a loading range of 0.3-0.6 mmol/g is considered optimal.
Protecting Groups and Coupling Strategies
1. Fmoc vs. Boc Strategy
Currently, the Fmoc/tBu strategy is the predominant approach in academic research due to its mild reaction conditions, compatibility with automated synthesis platforms, and relatively low incidence of side reactions.
2. Selection of Coupling Reagents
Highly efficient coupling reagents can substantially improve synthesis success rates. Commonly used reagents include:
For difficult sequences, the HATU + DIPEA system or a double coupling protocol is generally recommended.
3. Control of Deprotection Conditions
Fmoc deprotection is typically carried out using 20% piperidine under standard conditions. However, prolonged reaction times or elevated temperatures may induce undesirable side reactions, including:
Therefore, the deprotection step should be strictly controlled, typically 2 × 10 minutes.
Reaction Conditions and Instrument Parameters
1. Solvent Quality
The solvent quality must be carefully controlled to ensure that it is both amine-free and anhydrous. Excess moisture significantly reduces activation efficiency.
2. Reaction Temperature
3. Mixing and Agitation Efficiency
The mixing mechanism used in automated peptide synthesizers - such as mechanical agitation or nitrogen bubbling - directly affects coupling uniformity. Insufficient mixing can lead to incomplete or misleading coupling results.
Cleavage and Purification
1. Cleavage System
A typical TFA cleavage cocktail consists of:
For peptides containing Trp or Cys residues, ethanedithiol (EDT) is often added to prevent oxidative side reactions.
2. Crude Peptide Purity and HPLC Purification
Following cleavage, crude peptides are typically purified using reverse-phase high-performance liquid chromatography (RP-HPLC). Poor synthesis quality at earlier stages may result in:
Analytical Characterization: Ensuring Synthetic Quality
The success of SPPS depends not only on the synthesis process itself but also on accurate structural confirmation using high-resolution analytical techniques.
Common analytical methods include:
High-resolution mass spectrometry enables precise identification of:
Practical Strategies for Improving the Success Rate of SPPS
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
How to order?







