Targeted O-Glycosylation Site Analysis Service
MtoZ Biolabs offers Targeted O-Glycosylation Site Analysis Service for accurate identification and quantification of O-linked glycosylation sites on specific target proteins. This service is intended for clients who have defined target proteins (such as MUC1, PD-L1, EGFR, or α-Synuclein) and need to determine their O-glycosylation sites, glycan structures, and occupancy levels.
Using high-resolution Thermo Fisher Q Exactive HF and Orbitrap Fusion Lumos mass spectrometers coupled with Nano-LC, MtoZ Biolabs delivers high-quality, reproducible analytical results that support biopharmaceutical development, disease mechanism studies, and functional proteomics research.
What Is O-Glycosylation?
O-glycosylation is a key post-translational modification in which glycans are covalently attached to serine (Ser) or threonine (Thr) residues through an O-glycosidic bond. Unlike N-glycosylation, this modification lacks a defined consensus sequence and exhibits extensive microheterogeneity across tissues and cell types.
O-glycosylation plays fundamental biological roles in:
Protein Stability and Solubility: Preventing aggregation and enhancing protease resistance.
Cell Communication: Mediating receptor–ligand interactions and cell–cell adhesion.
Immune Modulation: Regulating antibody recognition and immune cell signaling.
Cancer Progression: Contributing to tumor invasion, metastasis, and immune evasion.
Therapeutic Development: Affecting pharmacokinetics and immunogenicity of biotherapeutics.
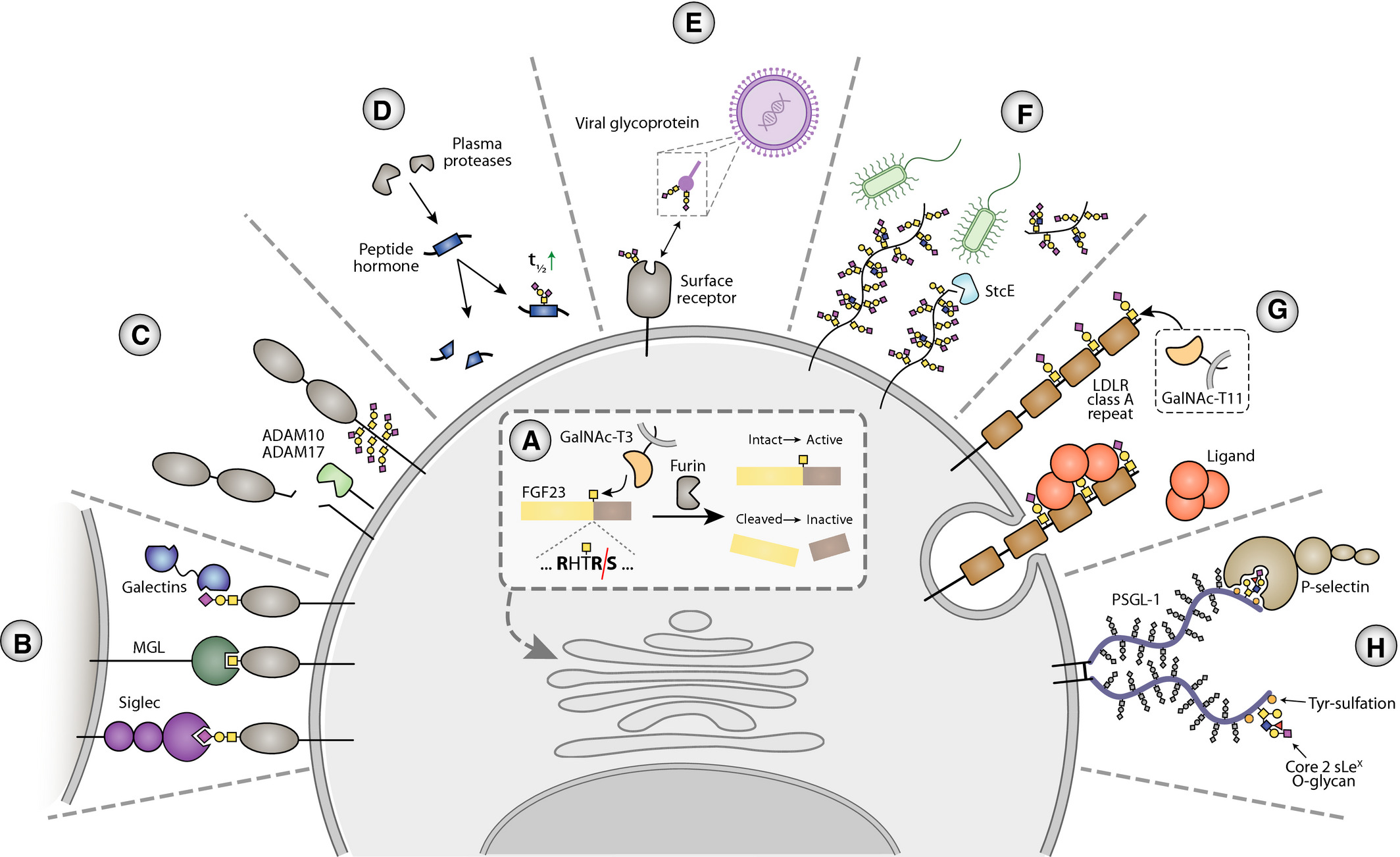
Wandall, H. H. et al. FEBS J. 2021.
Figure 1. Biological functions of O-Glycosylation
Targeted O-Glycosylation Site Analysis Service at MtoZ Biolabs
Targeted O-Glycosylation Site Analysis Service at MtoZ Biolabs focuses on precise site mapping for specific target proteins, employing two complementary analytical workflows for accurate site localization and glycan characterization.
1. β-Elimination Based Labeling Method (BEM)
In this workflow, O-linked glycans are selectively released through controlled β-elimination under non-reducing conditions, leaving specific mass tags on modified residues. The resulting β-eliminated peptides are then subjected to LC-MS/MS detection to determine O-glycosylation sites and differentiate them from phosphorylation sites.
This approach offers efficient large-scale screening of complex glycoprotein mixtures, making it particularly suitable for site-focused quantitative studies.
2. Intact Glycopeptide Analysis
The intact glycopeptide strategy preserves the original glycan-peptide linkage, enabling simultaneous identification of both the glycan structure and its corresponding attachment site. After enzymatic digestion and ZIC-HILIC enrichment, intact glycopeptides are separated via Nano-LC and analyzed using mass spectrometers.
This method provides deeper insights into site-specific glycan microheterogeneity, including sialylation and branching patterns.
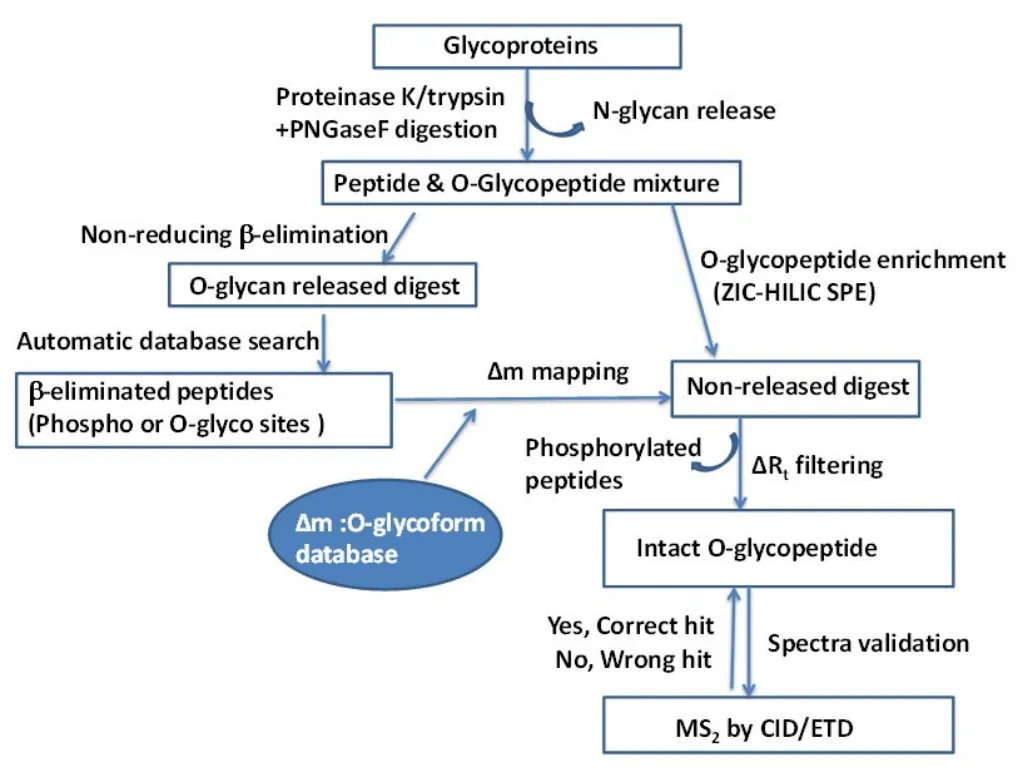
Huang, L, J. et al. J. Chromatogr. A. 2014.
Figure 2. Workflow of Targeted O-Glycosylation Site Analysis Service
Both methods are executed under strict analytical control using high-resolution LC-MS/MS, ensuring accuracy, reproducibility, and interpretability.
Why Choose MtoZ Biolabs?
✅ Comprehensive Dual-Strategy Platform: Integration of enzymatic and chemical methods ensures both precise site localization and structural characterization.
✅ High-Resolution MS Instrumentation: Orbitrap Fusion Lumos and Q Exactive HF platforms deliver superior mass accuracy and reproducibility.
✅ Broad Sample Compatibility: Applicable to purified proteins, recombinant therapeutics, serum, plasma, and complex cell lysates.
✅ Expert Scientific Support: Our experienced glycoproteomics specialists provide customized analysis design and professional data interpretation.
✅ One-Time-Charge: Our pricing is transparent, no hidden fees or additional costs.
Sample Submission Guidelines
MtoZ Biolabs accepts diverse target proteins for O-glycosylation site analysis. You can submit either purified proteins or biological samples enriched for the specific protein of interest.
|
Sample Type |
Recommended Amount |
|
Purified Proteins |
≥ 50 µg |
|
Recombinant Proteins |
≥ 50 µg |
|
Glycoprotein Mixtures |
≥ 100 µg |
|
Serum or Plasma |
≥ 100 µL |
|
Cell Lysates |
≥ 500 µg |
· Store samples at −80 °C and ship on dry ice in sealed containers.
· Avoid buffers containing detergents, salts, or denaturants that may inhibit enzymatic digestion.
· For low-abundance proteins, lectin affinity (e.g., Jacalin, VVA) or immunoprecipitation is recommended before LC–MS/MS.
· Provide the protein name, molecular weight, and any known or predicted O-glycosylation regions when available.
For more information, please refer to Sample Submission Guidelines for Proteomics.
Applications of Targeted O-Glycosylation Site Analysis Service
· Biopharmaceutical Characterization: Ensuring batch-to-batch glycoform consistency in therapeutic proteins and vaccines.
· Protein Engineering: Evaluating the impact of expression systems or site mutations on O-glycosylation occupancy.
· Disease Biomarker Discovery: Identifying O-glycosylation changes associated with cancer, autoimmune, and infectious diseases.
· Biosimilar and Vaccine Development: Comparing glycosylation profiles between originator biologics and biosimilar candidates.
· Functional Proteomics Studies: Exploring how O-glycosylation regulates protein folding, activity, and cellular communication.
Start Your Project with MtoZ Biolabs
Partner with MtoZ Biolabs to accelerate your glycoprotein research. Our Targeted O-Glycosylation Site Analysis Service delivers high-resolution, reproducible, and publication-ready data to support discovery, quality control, and therapeutic development.
Contact our technical specialists today to design your project or request a customized quotation.
Deliverables
1. Comprehensive Experimental Details
2. Materials, Instruments, and Methods
3. Total Ion Chromatogram & Quality Control Assessment
4. Data Analysis, Preprocessing, and Estimation
5. Bioinformatics Analysis
6. Raw Data Files
Related Services
How to order?







