Targeted N-Glycosylation Site Analysis Service
MtoZ Biolabs offers Targeted N-Glycosylation Site Analysis Service for precise identification and structural characterization of N-linked glycans on specific target proteins. This service is designed for clients who have identified one or more target proteins of interest (for example, IGF2BP3, EGFR, PD-L1, or Transferrin) and require detailed site-specific N-glycosylation information.
Using high-resolution Thermo Fisher Q Exactive HF and Orbitrap Fusion Lumos mass spectrometers combined with Nano-LC, MtoZ Biolabs provides accurate mapping of glycosylation sites, quantification of glycan occupancy, and comprehensive profiling of glycan structures to support biopharmaceutical characterization, functional proteomics, and disease-related glycoprotein studies.
What Is N-Glycosylation?
N-Glycosylation is a vital post-translational modification where complex oligosaccharides are covalently attached to asparagine residues within the Asn-X-Ser/Thr (NXS/T) motif. Each glycosylation site can host distinct glycan structures that modulate protein folding, solubility, stability, and biological activity.
Understanding the specific N-glycosylation sites on proteins is critical for:
Biopharmaceutical characterization, ensuring consistent glycoform patterns in recombinant proteins.
Disease mechanism studies, identifying altered glycosylation patterns in cancer, autoimmune, or metabolic diseases.
Protein engineering, evaluating how expression systems or mutations affect glycosylation efficiency.
Functional proteomics, correlating glycan occupancy with protein activity, trafficking, or signaling.
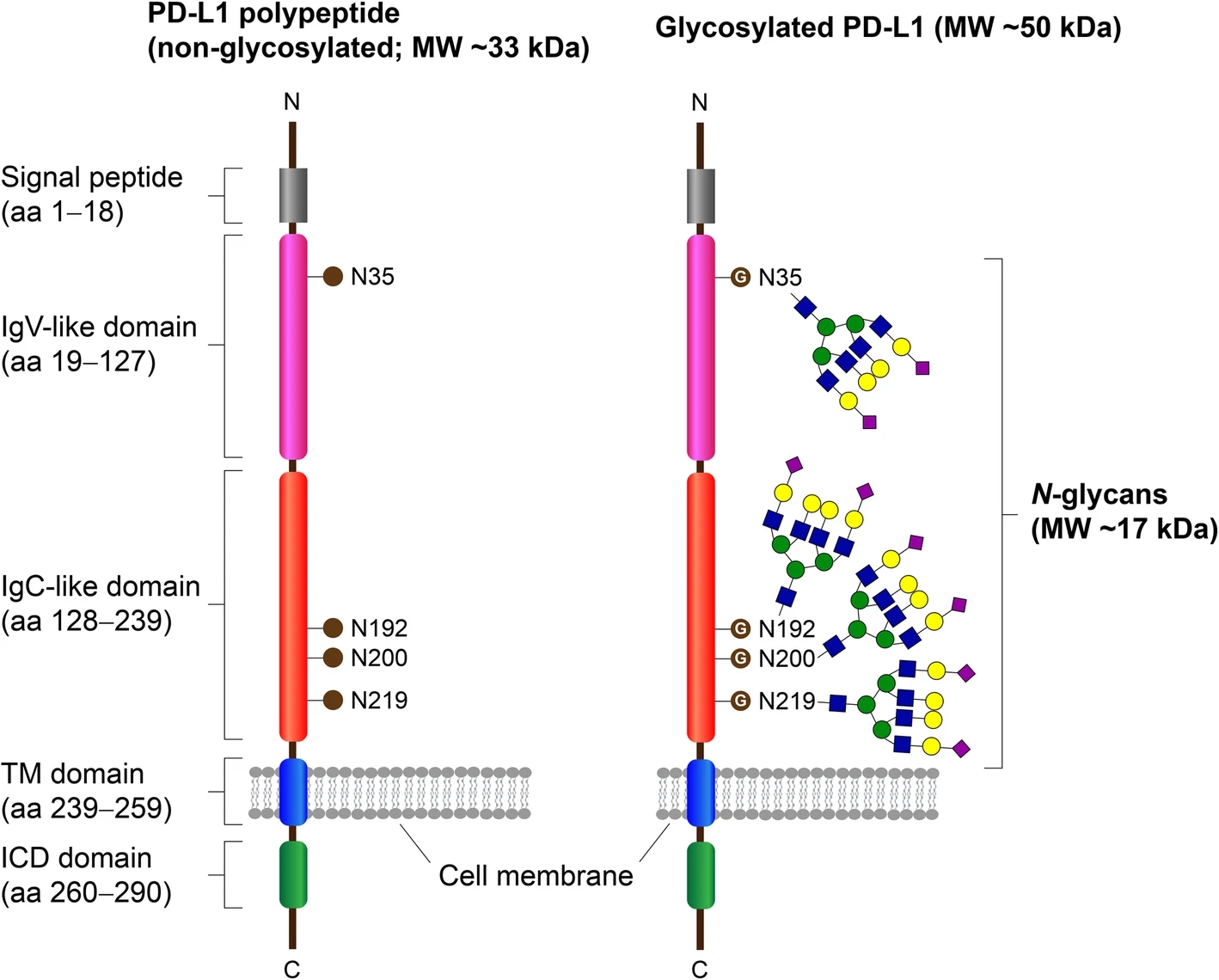
Wang, Y. N. et al. J. Biomed. Sci. 2020.
Figure 1. The Domain Structure and N-Glycosylation Sites on PD-L1
Targeted N-Glycosylation Site Analysis Service at MtoZ Biolabs
MtoZ Biolabs performs targeted N-glycosylation analysis using two complementary strategies optimized for predefined target proteins. These methods are suitable for both purified proteins and enriched target proteins from biological samples.
1. Heavy Water Glycan Release Method (H₂¹⁸O Labeling)
This approach identifies N-glycosylation sites through enzymatic deglycosylation performed in a heavy water (H₂¹⁸O) environment. During PNGase F digestion, an oxygen-18 isotope is incorporated at the cleavage site, resulting in a predictable +3 Da mass shift detectable by LC-MS/MS.
This method enables unambiguous site localization and quantification of glycan occupancy, making it particularly suitable for purified proteins and moderately complex biological samples.
2. Intact Glycopeptide Method
The intact glycopeptide approach preserves native glycan-peptide linkages, allowing simultaneous analysis of peptide sequences and attached glycans. After enzymatic digestion and HILIC-based enrichment, intact glycopeptides are separated by Nano-LC and analyzed with high-resolution Orbitrap MS systems.
This workflow provides detailed information on glycan composition, branching, and microheterogeneity, revealing how site-specific glycosylation affects protein structure and biological function.
Together, these two analytical strategies provide a complete understanding of N-glycosylation sites, combining precise isotopic localization with comprehensive glycan structural information, and ensuring reliable and reproducible results.
Application of Targeted N-Glycosylation Site Analysis Service
MtoZ Biolabs' Targeted N-Glycosylation Site Analysis Service is widely used in research and biopharmaceutical industries to explore site-specific glycosylation and its biological significance.
● Biopharmaceutical Characterization: Ensuring glycoform consistency and structural integrity in therapeutic proteins, monoclonal antibodies, and fusion proteins.
● Protein Engineering and Expression Optimization: Comparing glycosylation efficiency and site occupancy across different expression systems or engineered variants.
● Disease Mechanism Studies: Detecting abnormal glycosylation associated with cancers, immune diseases, and metabolic disorders.
● Functional Proteomics Research: Investigating how site-specific glycosylation influences protein stability, folding, and biological activity.
Why Choose MtoZ Biolabs?
✅ Advanced Analysis Platform: MtoZ Biolabs established an advanced protein PTM analysis platform, guaranteeing reliable, fast, and highly accurate analysis service.
✅ Dual-Approach Analytical Design: Combining isotope-labeled deglycosylation and intact glycopeptide mapping allows both precise site localization and in-depth glycan structure elucidation.
✅ Wide Sample Compatibility: Suitable for purified and recombinant proteins, plasma, serum, and cell lysates, enabling versatile applications in biological research and biopharmaceutical analysis.
✅ Expert Scientific Support: Our experienced scientists design customized analytical strategies, interpret data with precision, and provide technical consultation to ensure meaningful and actionable results.
✅ One-Time-Charge: Our pricing is transparent, no hidden fees or additional costs.
FAQ
Q1: What is the service general workflow?
Q2: What types of samples are suitable?
Our Targeted N-Glycosylation Site Analysis Service supportsboth purified proteins and biological samples containing predefined target proteins such as IGF2BP3, EGFR, PD-L1, or Transferrin. For best results, we recommend purified or well-characterized protein samples. If your sample is complex, our team can apply targeted LC-MS/MS or affinity enrichment to achieve accurate site-specific N-glycosylation profiling.
Q3: What data formats are provided?
We deliver both raw and processed LC-MS/MS data along with comprehensive analytical reports. Typical deliverables include:
Raw data files from Orbitrap LC-MS/MS systems
Annotated identification tables listing verified N-glycosylation sites, glycan compositions, and confidence scores
Quantitative summaries showing site occupancy or glycoform distribution when applicable
PDF summary report detailing the workflow, parameters, and key analytical results
All data tables are provided in Excel or CSV formats, ensuring compatibility with downstream bioinformatics and documentation systems.
Q4: How should I prepare my samples?
To ensure reliable results:
Avoid detergents, salts, and chaotropic reagents that may interfere with enzymatic digestion or ionization.
Clarify liquid samples by centrifugation before freezing.
Store all materials at −80 °C and ship on dry ice in sealed containers.
For targeted analysis, please indicate the protein name and expected glycosylation sites (Asn-X-Ser/Thr motifs) if available. For more information, please refer to Sample Submission Guidelines for Proteomics and Sample Submission Guidelines for Metabolomics.
Start Your Project with MtoZ Biolabs
Contact us to discuss your project or request a quotation. Whether you are evaluating glycosylation patterns in therapeutic proteins, investigating disease-related glycan alterations, or optimizing expression systems,
Related Services
How to order?







