Pullulan Analysis Service | Pharmaceutical Excipient
Pullulan is a water-soluble extracellular polysaccharide produced by fermentation of Aureobasidium pullulans. Chemically, it is a linear polysaccharide composed of maltotriose units linked by α-1,6-glycosidic bonds. Its unique molecular structure gives it good water solubility, film-forming ability, and low viscosity, and it is therefore widely used in the pharmaceutical, food, and healthcare industries.
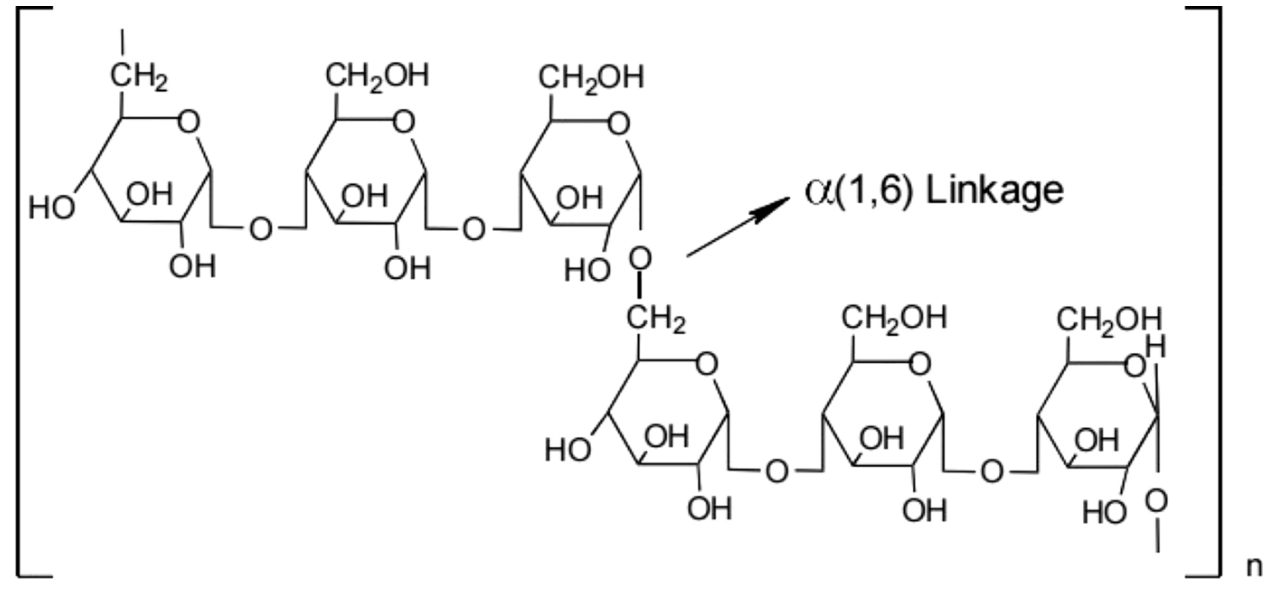
Aquinas, N. et al. Polym Bull. 2024.
Figure 1. The Strucuture of Pullulan
In the pharmaceutical field, Pullulan has multiple important functions as an excipient. It is often used in capsule shells, film coatings, sustained-release systems, and stabilizers. It can improve the appearance of drugs and patient compliance, and to some extent regulate drug release. At the same time, the natural origin and biocompatibility of Pullulan have made it a focus of regulatory and industry attention in recent years. Since the quality of excipients directly affects the safety and efficacy of drugs, systematic and scientific quality testing and characterization of Pullulan have become indispensable in drug development and production.
As a professional pharmaceutical excipient analysis and testing CRO, MtoZ Biolabs, relying on advanced mass spectrometry, chromatography, and spectroscopy platforms combined with the rich experience of a multidisciplinary team, provides comprehensive and standardized Pullulan Analysis Service that supports clients from qualitative identification, quantitative testing, and molecular weight distribution to structural and stability evaluation, delivering reliable data for R&D and production to ensure excipients comply with pharmacopeia and international standards.
Services at MtoZ Biolabs
MtoZ Biolabs’ pullulan analysis service covers multiple levels from basic identification to performance studies, ensuring that the identity, content, molecular characteristics, and application performance of samples are comprehensively evaluated.
| Qualitative Analysis | Thin-layer chromatography (TLC) combined with Pullulanase enzymatic hydrolysis is used to compare samples with standards. If the color development of the sample and the standard appears at the same position, the sample is confirmed as Pullulan polysaccharide. This method is direct, fast, and suitable for preliminary identification. |
| Quantitative Analysis | The anthrone-sulfuric acid method is used. By comparing with a standard curve, the content and purity of Pullulan in the sample are calculated. This method is highly sensitive, simple to operate, and suitable for routine determination. |
| Molecular Weight Determination | Size exclusion chromatography (SEC, also known as gel permeation chromatography) is employed to obtain the molecular weight distribution of Pullulan and calculate weight-average molecular weight (Mw), number-average molecular weight (Mn), and Z-average molecular weight (Mz). Molecular weight and its distribution are key parameters reflecting Pullulan’s physical properties and applicability. |
| Structural Characterization | Fourier transform infrared spectroscopy (FT-IR) is used to detect the chemical composition and structural features of Pullulan, identify functional groups, and evaluate sample purity. This method helps verify the structural consistency of Pullulan. |
| Stability Analysis | Thermogravimetric analysis (TGA) is used to study the thermal stability and decomposition behavior of Pullulan under different temperatures. By controlling the heating rate, the thermal degradation curve of Pullulan is obtained, which helps determine its storage and application conditions. |
| Rheological Properties | A rotational viscometer is used to measure the apparent viscosity of Pullulan solutions at different concentrations and shear rates. Mathematical models (such as the power law model) are applied to calculate flow consistency and behavior indices, revealing the rheological properties of Pullulan in formulations. |
| Routine Testing | Including loss on drying, ignition residue, heavy metal content, and microbial limits, ensuring sample safety and compliance with pharmacopeia requirements. |
| Capsule Performance Testing | For Pullulan capsules, elasticity, brittleness, and disintegration time can be evaluated, providing data to support the stability and safety of capsule dosage forms. |
Analysis Workflow
1. Sample Receipt and Registration: Confirm batch number, origin, and testing requirements.
2. Sample Pretreatment: Select dilution, filtration, or digestion steps according to the analysis project.
3. Experimental Testing: Conduct qualitative, quantitative, molecular weight, structural, stability, and rheological analysis.
4. Data Integration and Review: Ensure accuracy and comparability using standards and quality control samples.
5. Report Delivery: Generate a complete analysis report including methods, results, and spectra.
Service Advantages
1. Comprehensive Project Coverage
MtoZ Biolabs provides one-stop pullulan analysis service covering all key indicators, helping clients build a complete excipient quality profile.
2. Scientific and Standardized Methodology
Our analysis is based on established experimental methods and commonly used instrument platforms, balancing sensitivity and operability, ensuring data meet pharmacopeia standards, and facilitating regulatory submission and batch comparison.
3. Traceable and Reliable Results
Through strict quality control and standard material comparison, each test result is traceable and verifiable, providing clients with dependable scientific evidence.
4. Flexible Customization and Support
According to the client’s project requirements, we can flexibly combine testing items and report content to adapt to different stages such as R&D, production control, or regulatory submission, improving efficiency and data utilization.
Applications
✔️ Raw Material Release and Supplier Evaluation: Verify whether Pullulan quality meets batch consistency and regulatory requirements.
✔️ Formulation Development and Process Research: Provide physicochemical and rheological data for dosage form design and solution formulation.
✔️ Stability Studies: Evaluate Pullulan’s changes during storage and use.
✔️ Regulatory Submission and Compliance Testing: Provide data in line with regulatory requirements to support review and filing.
FAQs
Q1: How long does the Pullulan Analysis Service take?
A1: Routine analysis takes about 2–3 weeks, depending on sample quantity and project combination.
Sample Submission Suggestions
1. Sample Type: Both liquid and powder are acceptable.
2. Recommended Amount: Not less than 5 mL for liquid, not less than 2 g for solid.
3. Storage and Transport: Seal and protect from light. Short-distance transport at room temperature is acceptable; please specify if special conditions are required.
4. Additional Information: Provide batch number, origin, and testing requirements.
Deliverables
1. Complete analysis report in PDF format
2. Raw data and processed tables
3. Spectra and quality control results
4. Comparative and trend analysis (if required)
5. Methodology and deviation explanation (if applicable)
As a pharmaceutical excipient, the quality control of Pullulan is directly related to the stability and safety of drugs. With advanced platforms and a professional team, MtoZ Biolabs provides systematic and reliable pullulan analysis service to help clients meet multi-stage requirements from R&D to submission. If you are looking for a professional excipient testing partner, please contact MtoZ Biolabs. We will safeguard your projects with scientific data and efficient service.
How to order?







