Peptide Drug Immunogenicity Testing Service
MtoZ Biolabs provides Peptide Drug Immunogenicity Testing Service dedicated to evaluating anti-drug antibodies generated in response to peptide therapeutics. ADA assessment is essential for understanding immune recognition, monitoring changes in drug performance, and supporting the overall safety evaluation of peptide-based drug candidates. With optimized immunoassay platforms and proven analytical expertise, MtoZ Biolabs delivers reliable datasets that help guide formulation refinement, dose selection, and risk assessment during development.
Overview
Peptide drugs, despite their relatively small size, can elicit immune responses when introduced into biological systems. These responses manifest as anti-drug antibodies that may alter pharmacokinetics, diminish efficacy, or interfere with the intended biological activity. ADA testing provides a structured approach to quantify and characterize these responses.
A comprehensive ADA assessment typically includes:
● Detection of potential immune responses through screening assays
● Confirmation of antibody specificity
● Quantitative titer determination to evaluate response magnitude
This framework helps researchers understand how peptide sequence, chemical modifications, aggregation, impurities, or delivery methods contribute to immunogenicity, enabling more informed decision-making during drug development.
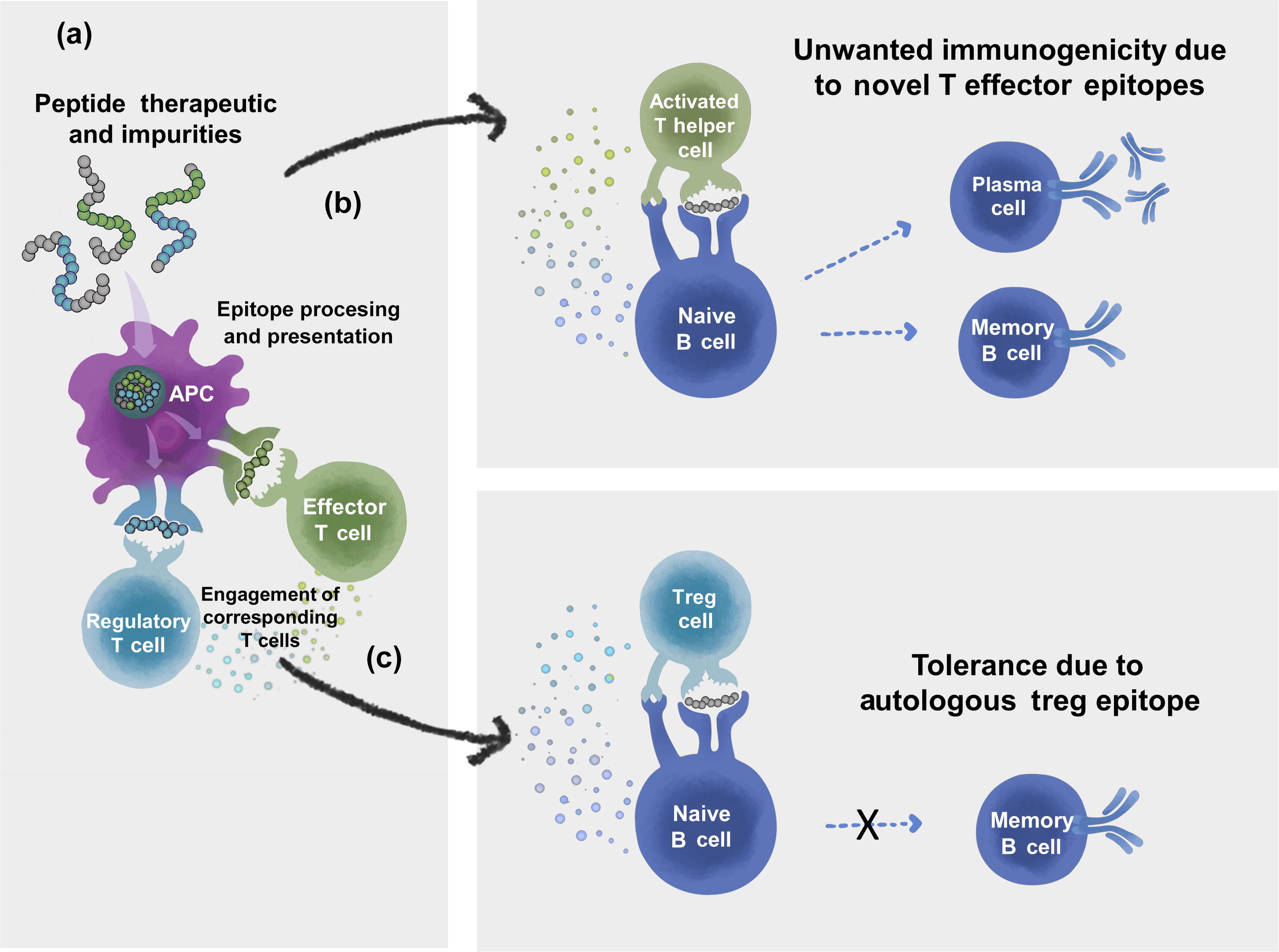
De Groot, A. S. et al. Drug Discov. Today. 2023.
Figure 1. Overview of Immune Pathways Activated by Peptide Therapeutics
Peptide Drug Immunogenicity Testing Service at MtoZ Biolabs
MtoZ Biolabs offers a complete ADA analysis workflow designed specifically for peptide drugs and peptide conjugates. Each step is performed using validated procedures and high-sensitivity immunoassay technologies.
1. ADA Screening Assay
Initial screening is performed using ELISA or electrochemiluminescence systems to identify ADA signals in serum or plasma samples with high sensitivity.
2. ADA Confirmatory Assay
Confirmatory testing evaluates the specificity of detected antibody binding using competitive inhibition strategies to distinguish true ADA responses from non-specific interactions.
3. ADA Titer Measurement
Quantitative titration is conducted to assess the immune response magnitude and support longitudinal or comparative studies across formulations, dosing regimens, or animal models.
All assays include appropriate controls, cut point evaluation, and optimized parameters to ensure data reproducibility and scientific reliability.
Why Choose MtoZ Biolabs?
✔ Specialized ADA Testing Expertise: Experienced scientists skilled in designing and executing ADA assays tailored to the characteristics of peptide therapeutics.
✔ Advanced Immunoassay Platforms: High-sensitivity ELISA and electrochemiluminescence systems allow accurate detection of low-level ADA responses in complex biological matrices.
✔ Reproducible and Reliable Data: Standardized procedures, validated workflows, and stringent documentation ensure consistent and traceable results.
✔ Integrated Analytical Options: ADA testing can be complemented with peptide stability assessment, endotoxin analysis, and product characterization to support comprehensive quality evaluation.
✔ Dedicated Scientific Support: Consultation on assay design, study planning, and data interpretation ensures smooth project execution and well-supported conclusions.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs offers high-quality Peptide Drug Immunogenicity Testing Service to support the development, evaluation, and refinement of peptide therapeutics. Contact our team to discuss your project needs and analytical strategy.
FAQ
Q1: What types of samples are suitable?
Peptide drugs, formulated peptides, and serum or plasma samples collected from immunogenicity studies.
Q2: What is the service general workflow?
Q3: What data formats are provided?
MtoZ Biolabs provides comprehensive documentation and data outputs, including:
a full PDF report detailing methods, conditions, and analytical results
screening, confirmatory, and titer assay data
calibration and QC performance metrics
interpretation notes to support downstream decision-making
spreadsheet files containing raw absorbance or signal values upon request
These deliverables facilitate seamless integration into internal records, project reports, or regulatory submissions.
Q4: How should I prepare my samples?
To ensure high-quality ADA analysis:
provide sterile samples without detergents or additives
use low-binding tubes for peptide materials
store samples refrigerated or frozen depending on peptide stability
avoid repeated freeze-thaw cycles
include detailed information on concentration, formulation, and handling
If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







