Peptide Drug Biochemical Analysis Service
MtoZ Biolabs provides comprehensive Peptide Drug Biochemical Analysis Service designed to support the characterization, optimization, and functional evaluation of peptide-based therapeutics. Our analytical platform offers precise, high-quality biochemical data that help researchers understand peptide activity, stability, binding behavior, and overall developability across discovery and preclinical stages.
Overview
Biochemical analysis is essential for understanding how peptide drug molecules behave in biological systems. Peptide therapeutics participate in diverse biochemical processes, including enzymatic reactions, receptor engagement, signaling modulation, and protein complex formation. Systematic biochemical assays enable researchers to measure molecular interactions, quantify functional responses, track biochemical activity over time, and identify performance liabilities that may influence drug development.
Typical biochemical studies may include enzymatic reaction monitoring, activity measurements, peptide-protein binding evaluation, immunoassay-based quantitation, and solubility or aggregation assessments. These analyses help define potency, confirm mechanism of action, assess biochemical stability, and determine how peptide modifications influence function. MtoZ Biolabs integrates biochemical expertise with state-of-the-art analytical technologies to provide Peptide Drug Biochemical Analysis Service that informs peptide engineering, formulation optimization, and development decision-making.
Peptide Drug Biochemical Analysis Service at MtoZ Biolabs
MtoZ Biolabs offers a focused suite of biochemical assays and analytical evaluations tailored to the unique requirements of peptide drug development.
1. Enzymatic and Functional Activity Assays
Biochemical evaluation of enzyme-mediated reactions, catalytic turnover, and peptide-induced modulation of enzymatic pathways.
2. Immunoassay-Based Quantification
Antibody-driven assays such as ELISA for sensitive measurement of peptide concentration, activity changes, or target binding effects.
3. Receptor and Protein Binding Studies
Characterization of peptide interactions with receptors, target proteins, or biomolecular partners to support potency and mechanism analysis.
4. Solubility, Aggregation, and Physical Property Assessment
Evaluation of physicochemical parameters such as solubility, aggregation tendency, and structural stability under formulation or physiological conditions.
5. Biochemical Stability and Degradation Analysis
Assessment of peptide integrity under enzymatic, thermal, oxidative, or pH-related stresses to identify key degradation pathways.
Why Choose MtoZ Biolabs?
✅ Comprehensive Biochemical Capabilities
Extensive assay options covering enzymatic activity, binding interactions, immunoassays, and functional biochemical measurements.
✅ Advanced Analysis Platform
High-quality instrumentation and validated assay formats ensure accurate, reproducible biochemical results.
✅ Experienced Scientific Team
Expert guidance in assay selection, study setup, troubleshooting, and interpretation of complex biochemical data.
✅ Flexible Study Design
Customizable workflows tailored to peptide class, research objectives, and project timelines.
✅ One-Time-Charge
Our pricing is transparent, no hidden fees or additional costs.
Start Your Project with MtoZ Biolabs
Advance your peptide drug development with accurate, comprehensive biochemical analysis tailored to your research goals. MtoZ Biolabs delivers high-quality data that support mechanism studies, stability evaluation, potency assessment, and candidate optimization.
FAQ
Q1: What types of samples are suitable?
We analyze linear peptides, cyclic peptides, stapled peptides, conjugated peptides, peptidomimetics, peptide inhibitors, and other engineered peptide formats. Both purified materials and formulated samples are accepted.
Q2: What is the service general workflow?
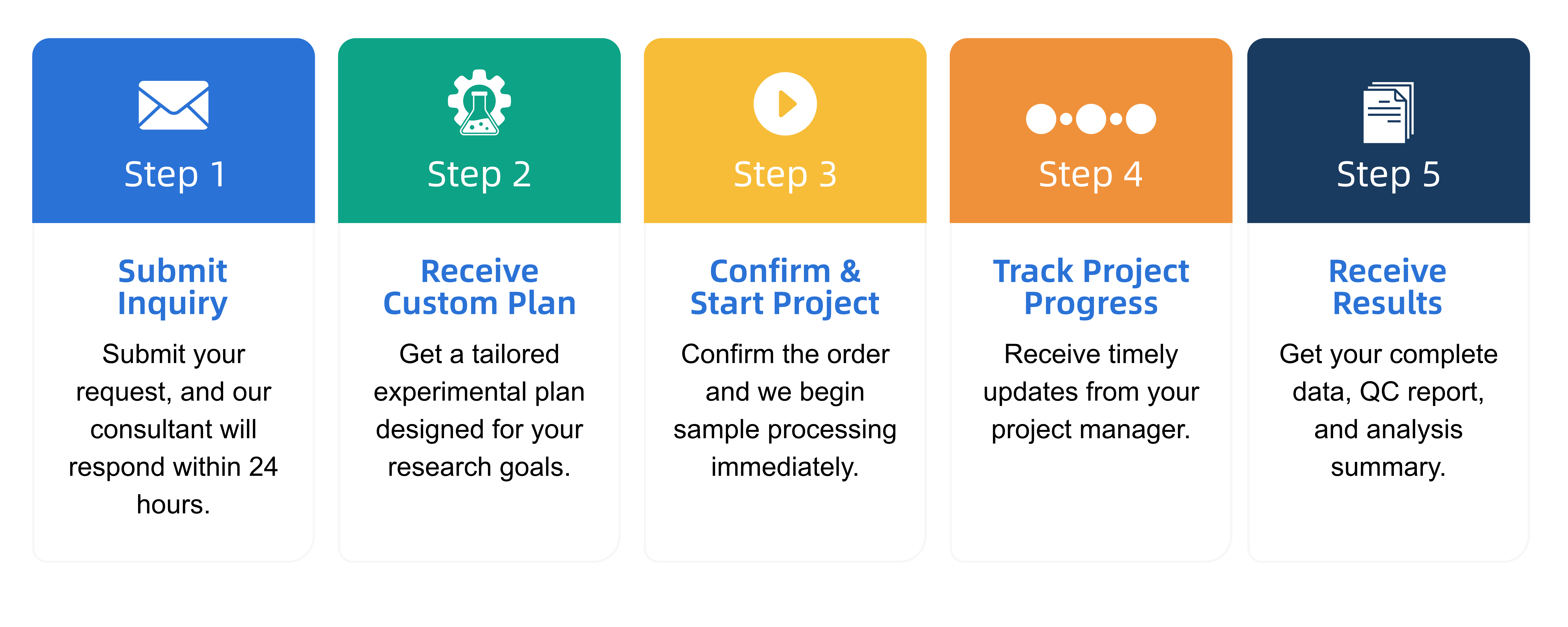
Q3: What data formats are provided?
We provide raw analytical files, processed datasets in Excel or CSV format, annotated chromatograms or spectra in PDF, graphical biochemical outputs, method documentation, and a complete interpretive report summarizing all findings.
Q4: How should I prepare my samples?
To ensure high-quality results, follow these guidelines:
Store samples at -80℃ and avoid repeated freeze-thaw cycles
Provide sufficient peptide quantity and volume for planned assays
Supply full details on buffers, solvents, salts, and stabilizers
Clarify samples by centrifugation if particulates are present
Ship samples on dry ice in insulated containers
For more information, please refer to Sample Submission Guidelines for Proteomics. If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







