Peptide Drug Bacterial Endotoxin Testing Service
MtoZ Biolabs provides specialized Peptide Drug Bacterial Endotoxin Testing Service for evaluating and controlling endotoxin contamination in peptide-based therapeutics and formulations. Trace endotoxin residues can interfere with experimental reproducibility, trigger immune activation, and reduce cell viability in culture. Our Peptide Drug Bacterial Endotoxin Testing Service ensures that peptide products meet stringent cleanliness and quality requirements, enabling consistent biological performance and reliable analytical outcomes. Using high-sensitivity Limulus Amebocyte Lysate (LAL) assays optimized for peptide matrices, MtoZ Biolabs delivers accurate and reproducible endotoxin quantification for research, manufacturing, and preclinical applications.
Overview
Endotoxins, also known as lipopolysaccharides (LPS), are integral components of Gram-negative bacterial cell walls. During peptide synthesis, purification, or formulation, trace endotoxins may inadvertently enter the product, posing a risk to in vitro and in vivo studies. Even minimal contamination can trigger immune responses, inhibit transfection efficiency, and distort experimental results.
Peptide drug development requires stringent endotoxin control to safeguard cell culture performance, maintain assay consistency, and ensure product quality. MtoZ Biolabs applies matrix-compatible detection methods and rigorous quality control procedures to quantify endotoxin levels with precision. This ensures that peptide formulations meet the strict cleanliness requirements necessary for downstream biological evaluation.
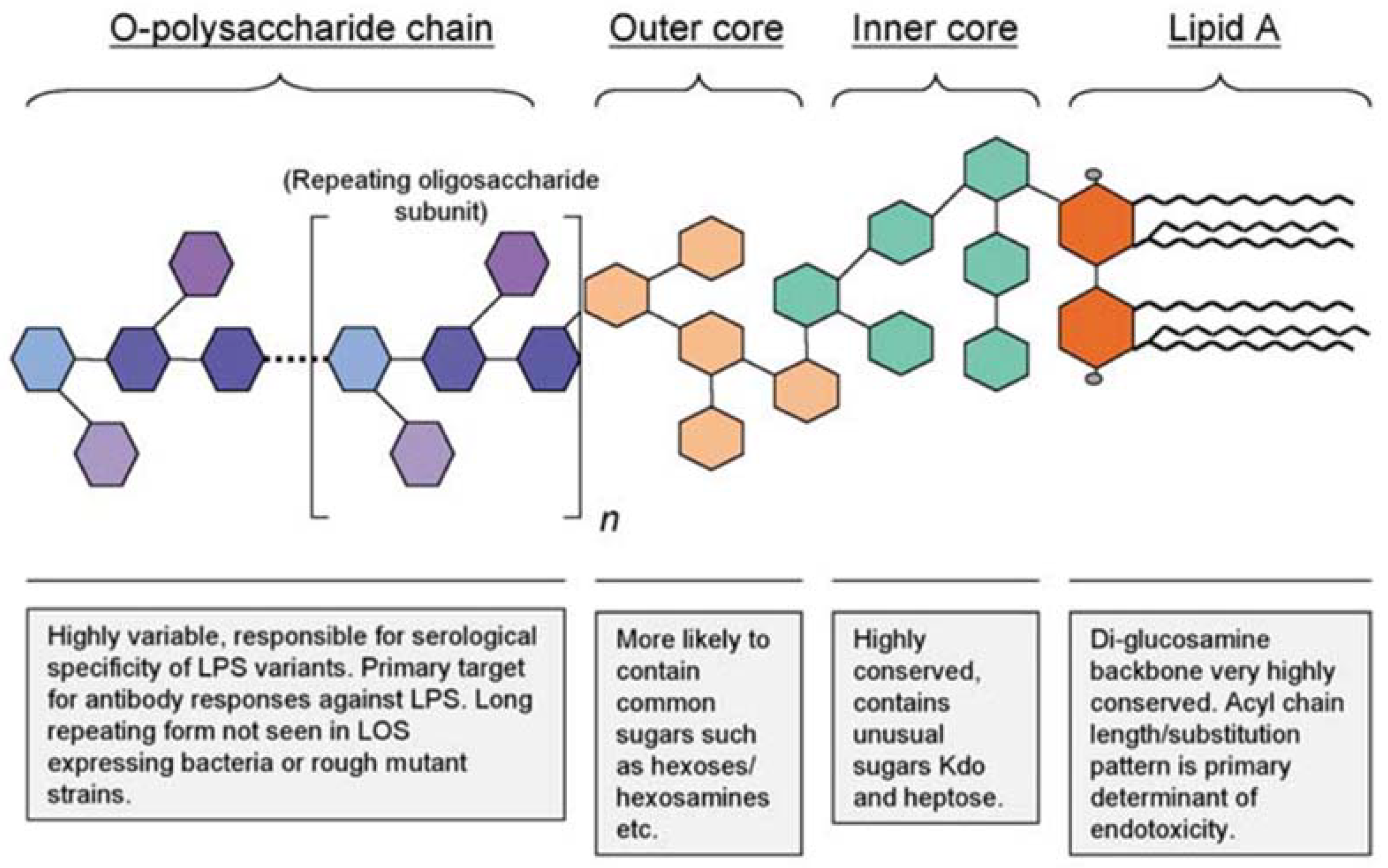
Eckel, E. F. et al. J. Dairy Sci. 2020.
Figure 1. The General Structure of Gram-Negative Bacterial Endotoxins
Peptide Drug Bacterial Endotoxin Testing Service at MtoZ Biolabs
MtoZ Biolabs provides a comprehensive portfolio of bacterial endotoxin testing solutions for peptide-based products. Our service offerings are designed to address diverse analytical requirements in peptide synthesis, purification, and formulation quality control.
1. Qualitative Endotoxin Detection
Rapid identification of endotoxin presence using gel-clot Limulus Amebocyte Lysate (LAL) assays to verify contamination status.
2. Quantitative Endotoxin Measurement
Precise determination of endotoxin concentration through kinetic turbidimetric or chromogenic LAL assays, ensuring reliable quantitative evaluation.
3. Comparative Endotoxin Evaluation
Assessment of endotoxin levels across different production or purification stages to support process validation and contamination control.
4. Matrix Compatibility Testing
Evaluation of peptide matrix interference to ensure analytical accuracy and assay recovery consistency under complex formulation conditions.
5. Extended Analytical Integration
Optional combination with peptide purity, stability, or sequence analysis to support multidimensional product quality assessment.
Why Choose MtoZ Biolabs?
✅ Validated Analytical Expertise: Extensive experience in bacterial endotoxin detection for peptides, peptide-drug conjugates, and related biologics.
✅ Matrix-Specific Optimization: Customized assay conditions to accommodate hydrophobic, amphiphilic, or highly charged peptide formulations.
✅ Comprehensive Detection Capability: Multiple LAL assay formats covering qualitative screening and quantitative measurement needs.
✅ Strict Quality Management: All analyses are conducted under controlled laboratory environments with systematic quality assurance review.
✅ Integrated Analytical Solutions: Seamless integration with peptide characterization, purity testing, and stability analysis to ensure consistent data quality.
✅ Experienced Scientific Team: Professional analysts provide expert consultation for project design, data interpretation, and technical recommendations.
Applications of Peptide Drug Bacterial Endotoxin Testing Service
· Endotoxin detection for peptide and peptide-drug conjugates.
· In-process testing during peptide synthesis and purification.
· Quality control for peptide formulation and release studies.
· Assessment of endotoxin contamination in research-grade materials.
· Validation of depyrogenation efficiency in peptide production.
Start Your Project with MtoZ Biolabs
MtoZ Biolabs delivers accurate and reliable bacterial endotoxin testing specifically designed for peptide-based drugs and formulations. Our expert team ensures consistent analytical performance and supports your research and development needs with confidence. Contact us today to discuss your testing project or request a customized quotation.
FAQ
Q1: What types of samples are suitable?
MtoZ Biolabs accepts lyophilized peptides, peptide-drug conjugates, peptide formulations, and buffers for endotoxin testing. Custom sample types can be evaluated upon request.
Q2: What is the service general workflow?
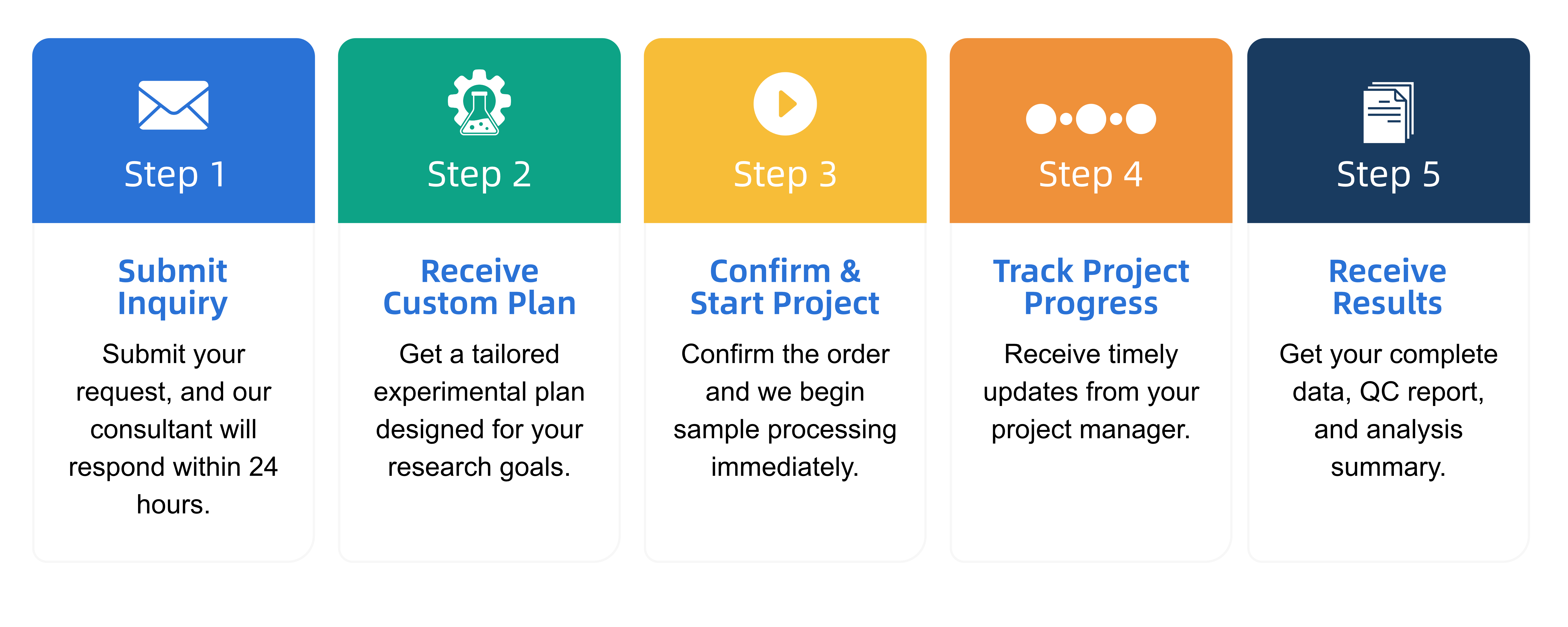
Q3: What data formats are provided?
Clients receive a detailed analytical report including raw data, calculated endotoxin concentrations, calibration curves, quality control recovery results, and interpretation notes in PDF format. Additional data formats can be provided if required.
Q4: How should I prepare my samples?
· Use sterile, endotoxin-free containers for collection and shipment.
· Avoid surfactants or organic solvents that may interfere with detection.
· Store samples at -20 °C and ship under refrigerated conditions.
· Provide detailed formulation information, including concentration and buffer composition.
For more information, please refer to Sample Submission Guidelines for Proteomics. If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
How to order?







