Lung Cancer-Targeted Exosome Modification Service
- Define targets, drug types, and delivery scenarios
- Recommend optimal modification strategies (e.g., chemical conjugation, gene engineering, membrane fusion)
- Isolate high-purity exosomes from designated cells (e.g., mesenchymal stem cells, tumor cells) via ultracentrifugation and size-exclusion techniques
- Characterization via NTA, TEM, and Western blot for exosome markers
- Surface Modification: Ligand or antibody conjugation
- Drug Loading: Encapsulation of small molecules, nucleic acids (siRNA/miRNA), or proteins via electroporation, sonication, or incubation
- Gene Engineering: Transfection or lentiviral delivery to express targeting proteins in parental cells
- Targeting Efficiency: Flow cytometry, confocal microscopy for cellular uptake
- Drug Release: Simulated physiological and tumor environments to assess sustained release profiles
- In Vitro Efficacy: MTT/EdU for proliferation inhibition, Annexin V for apoptosis, wound healing/Transwell for migration assays
Exosomes are nano-sized vesicles (30–150 nm in diameter) naturally secreted by cells and widely distributed in body fluids such as blood, saliva, and urine. These vesicles carry biologically active molecules including proteins, nucleic acids (mRNA, miRNA, lncRNA), and lipids, serving as vital mediators of intercellular communication. Due to their innate low immunogenicity, excellent biocompatibility, high stability, and ability to cross biological barriers, exosomes are considered ideal carriers for drug delivery. Through engineering modifications, exosomes can be endowed with precise targeting, efficient drug loading, and intelligent release capabilities, offering great potential in gene therapy, immunomodulation, and chemotherapeutic drug delivery.
Lung cancer remains one of the leading causes of cancer-related mortality worldwide. Traditional chemotherapy and targeted therapies often suffer from insufficient delivery efficiency, poor tumor specificity, and systemic toxicity. Leveraging the natural delivery potential of exosomes, engineered modifications can overcome these therapeutic limitations. Surface conjugation with lung cancer-specific ligands or antibodies enables precise recognition of tumor markers, significantly reducing off-target toxicity. Their nano-scale structure and penetrability allow them to cross the blood–brain barrier and dense tumor microenvironment (TME), delivering therapeutic molecules directly to the tumor core. Additionally, by encapsulating chemotherapeutic drugs or gene regulatory molecules (e.g., siRNA/miRNA), exosomes can enhance local drug concentration and modulate resistance gene expression—offering innovative strategies to overcome therapeutic resistance.
Chen, H. et al. Front Bioeng Biotechnol. 2023.
Figure 1. Workflow of Lung Cancer-Targeted Exosome Modification
Exosome Modification Strategies for Lung Targeting :
1. Ligand/Antibody Conjugation: Surface coupling of lung cancer-specific ligands or antibodies to enhance targeting ability.
2. Peptide Modification: Insertion of targeting peptides via gene engineering or chemical conjugation to bind lung-specific receptors.
3. Biomembrane Fusion: Fusion with natural membranes (e.g., macrophage membrane) with lung homing capability to retain innate tropism.
MtoZ Biolabs offers a comprehensive Lung Cancer-Targeted Exosome Modification Service for research institutions and pharmaceutical enterprises, covering targeted modification, drug loading, functional validation, and efficacy evaluation. Through modular design, we tailor modification strategies based on client needs, supporting innovative drug development for lung cancer.
Table 1. Lung Cancer-Targeted Exosome Modification Service at MtoZ Biolabs
| Research Needs | Our Services |
| Exosome Source Customization | Isolation from stem cells, tumor cells, plasma/serum |
| Targeted Modification | Ligand conjugation, antibody engineering, membrane fusion, peptide insertion |
| Drug Loading | siRNA/miRNA encapsulation, small-molecule chemotherapeutics, geneediting tools |
| Functional Validation | Cellular uptake, tumor spheroid penetration, in vivo biodistribution, efficacy/toxicity assessment |
| Translational Support | Scale-up, stability testing |
Analysis Workflow
1. Needs Assessment and Strategy Design
2. Exosome Preparation and Purification
3. Targeting Modification and Functionalization
4. Functional Validation and Optimization
5. Optional In Vivo Efficacy Evaluation
Establish lung cancer xenograft models and monitor targeted distribution and antitumor effects via in vivo imaging
Service Advantages
1. Precision Delivery Design
Multi-dimensional targeting (ligand + antibody + membrane fusion) significantly enhances lung accumulation efficiency (>80%)
2. Standardized Workflow
From isolation to validation, all steps follow ISO-compliant quality management systems to ensure reproducibility
3. Flexible Customization
Supports small- to mid-scale production (mg–g level) to meet requirements at various drug development stages
4. Stringent Quality Control
Triple QC for exosome purity, drug loading efficiency (HPLC quantification), and biological activity (cellular uptake)
5. Cross-Disciplinary Technical Support
Comprehensive pathology analysis, pharmacodynamic assessment, and mechanistic studies by a biomedical expert team
Applications
1. Chemotherapy Drug Targeted Delivery
Encapsulation of doxorubicin, paclitaxel, etc., to enhance tumor site concentration and reduce cardiotoxicity, hepatotoxicity, and nephrotoxicity
2. Immunomodulation and Combination Therapy
Delivery of PD-1/PD-L1 antibodies or CAR-T-related nucleic acids to reshape the immune microenvironment and boost immune checkpoint blockade efficacy
3. Gene Therapy and Resistance Reversal
Delivery of siRNA/miRNA to regulate tumor signaling pathways and reverse drug resistance
4. Diagnostics and Prognostic Monitoring
Enrichment of lung cancer biomarkers for liquid biopsy and therapeutic response evaluation
Case Study
1. Cancer Cell-Derived Exosome Based Dual-Targeted Drug Delivery System for Non-Small Cell Lung Cancer Therapy
This study constructs a "dual-targeting" drug delivery platform for non-small cell lung cancer (NSCLC) therapy by combining exosomes derived from NSCLC cells with superparamagnetic iron oxide nanoparticles (SPIONs). The system utilizes the homing ability of exosomes and the magnetic targeting ability of SPIONs to promote targeted delivery of doxorubicin (DOX) at the cellular level. In NCI-h1299 tumor models, Exo-SPIONs loaded with DOX demonstrated enhanced tumor tissue delivery, tumor suppression under an external magnetic field, and reduced toxicity to normal tissues. This "dual-targeting" drug delivery platform holds promise for targeted chemotherapy in NSCLC. Lung Cancer-Targeted Exosome Modification Service focuses on developing targeted exosome delivery platforms for lung cancer therapy. By engineering exosomes to enhance their tissue-homing abilities and combining multiple targeting strategies, we enable efficient drug enrichment and delivery. Our services include exosome modification, drug loading, and functional validation, suitable for various lung cancer research models.
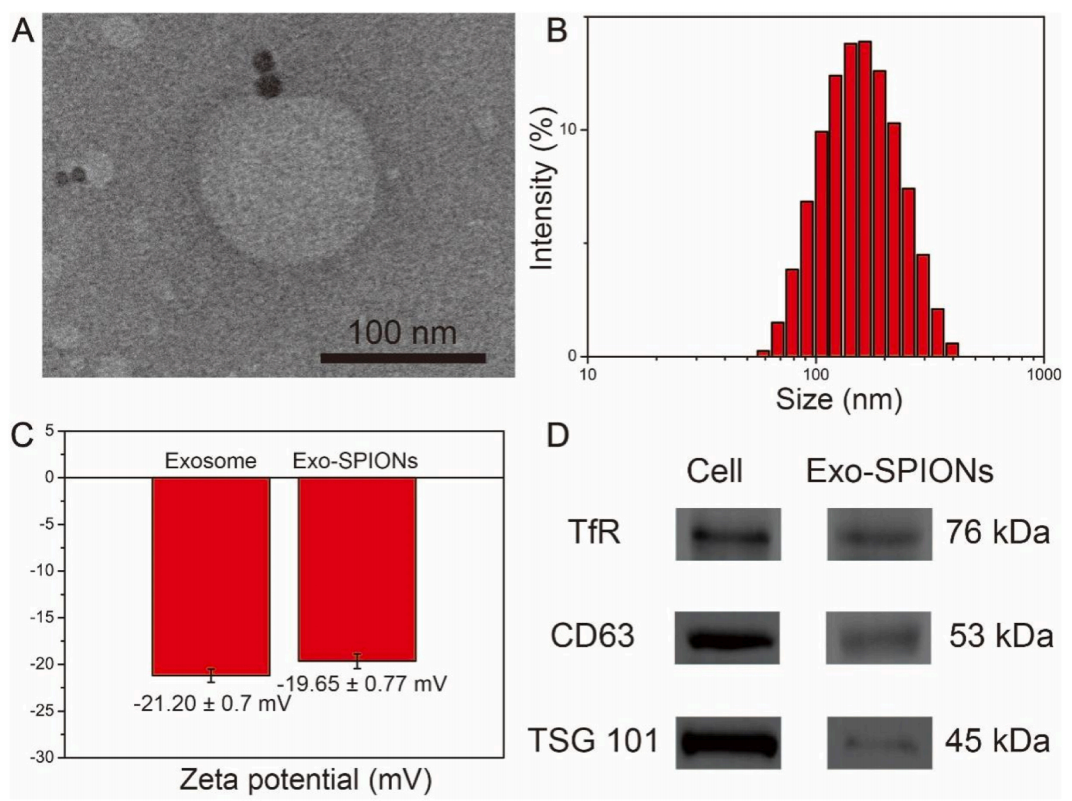
Wang, J. et al., Colloids Surf B Biointerfaces, 2024.
Figure 3. Characterization and Identification of Exo-SPIONs
2. Docetaxel-Loaded Exosomes for Targeting Non-Small Cell Lung Cancer: Preparation and Evaluation in vitro and in vivo
This study investigates the use of docetaxel-loaded exosomes (EXO-DTX) for targeting non-small cell lung cancer (NSCLC). Exosomes were isolated from A549 cancer cells and characterized by transmission electron microscopy, nanoparticle tracking analysis, and Western blotting. DTX was loaded into exosomes via electroporation. The in vitro evaluation demonstrated that EXO-DTX exhibited higher cytotoxicity, increased cellular uptake, and enhanced tumor targeting compared to free DTX. In vivo, EXO-DTX showed better targeting to tumor tissues, suggesting exosomes as a promising drug delivery system for NSCLC therapy. Lung Cancer-Targeted Exosome Modification Service offers advanced solutions for developing exosome-based drug delivery systems specifically targeting lung cancer. By utilizing exosomes derived from tumor cells, this service enhances drug delivery efficiency and targeting precision, providing a potential method for improving therapeutic outcomes in lung cancer treatment.
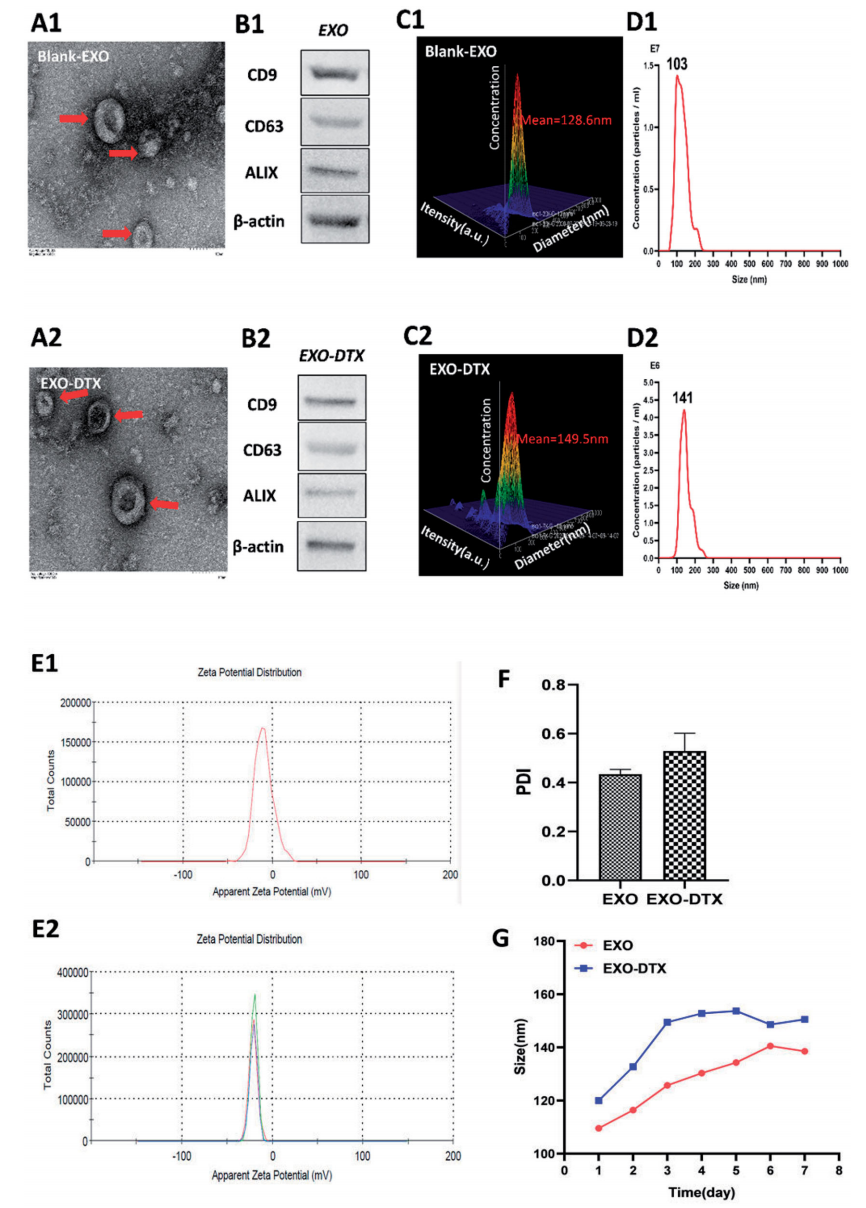
Wang, Y. et al., Drug Deliv, 2021.
Figure 4. Characterization of the EXO and EXO-DTX
Engineered exosomes offer a highly specific, low-toxicity, and long-circulating solution for lung cancer therapy. With extensive experience in exosome engineering, full-chain technical capabilities, and rigorous quality control systems, MtoZ Biolabs is committed to supporting your R&D and clinical translation needs from start to finish. Contact us today for customized strategy design and a free technical consultation!
How to order?







