Low-substituted Hydroxypropyl Cellulose Analysis Service | Pharmaceutical Excipient
- Sample Type: Powdered L-HPC
- Minimum Quantity: 5 grams
- Storage Conditions: Keep sealed and dry at room temperature
MtoZ Biolabs provides Low-Substituted Hydroxypropyl Cellulose Analysis Service to support comprehensive characterization and quality assessment of low-substituted hydroxypropyl cellulose (L-HPC), a widely used pharmaceutical excipient. L-HPC is a nonionic cellulose ether produced through the partial etherification of cellulose with propylene oxide under controlled conditions. Its porous morphology, large surface area, and excellent swelling properties make it an ideal disintegrant and binder in solid dosage formulations.
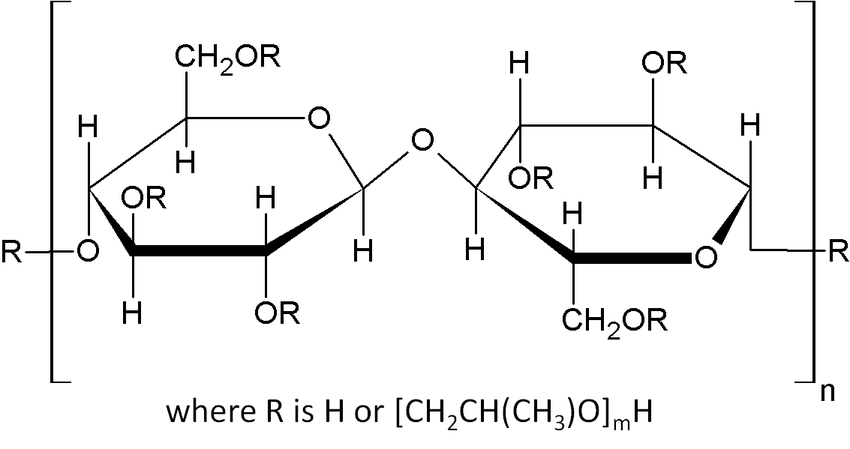
Figure 1. Molecular Structure of Low-substituted Hydroxypropyl Cellulose
The physicochemical characteristics of L-HPC, including its hydroxypropyl content, degree of substitution, moisture level, and viscosity, play a crucial role in determining tablet hardness, disintegration rate, and drug release performance. MtoZ Biolabs utilizes advanced analytical technologies and validated workflows to ensure reliable and reproducible results. Our Low-Substituted Hydroxypropyl Cellulose Analysis Service provides essential data that helps ensure batch consistency, optimize formulation design, and meet regulatory requirements.
Services at MtoZ Biolabs
MtoZ Biolabs offers a comprehensive suite of analytical solutions for the evaluation of L-HPC’s identity, purity, and performance-related parameters. Each analysis is conducted under strict quality control to ensure data accuracy and reliability.
● Identification
FTIR spectroscopy and colorimetric assays are used to confirm the identity of L-HPC. The appearance of a blue-green color with anthrone sulfate reagent and a white flocculent precipitate upon reaction with sodium hydroxide and an acetone-methanol mixture confirms the presence of L-HPC.
● Solubility Testing
L-HPC disperses in water to form a colloidal suspension but remains insoluble. It is also insoluble in ethanol, acetone, and ether, while dissolving in a 10 percent sodium hydroxide solution.
● Hydroxypropyl Content and Degree of Substitution
Gas chromatography with a phenyl methyl polysiloxane stationary phase or titration after distillation is used to quantify hydroxypropyl content. These results reflect the substitution degree that determines hydration behavior and compressibility.
● pH Measurement
The pH of a 1 percent aqueous dispersion is measured using a calibrated pH meter, typically between 5.0 and 7.5.
● Loss on Drying
Samples are dried under controlled conditions until constant weight. The loss on drying should not exceed 5 percent to ensure product stability.
● Heavy Metal and Arsenic Content
ICP-MS and colorimetric assays confirm that metal impurities and arsenic levels are within acceptable limits.
● Chloride Test
Chloride content is determined using silver nitrate titration, with comparison against a sodium chloride standard solution.
● Scorch Residue Test
After full incineration, the remaining ash should not exceed 1 percent, ensuring high purity.
Through these evaluations, MtoZ Biolabs’ Low-Substituted Hydroxypropyl Cellulose Analysis Service provides a full understanding of material composition and performance, supporting formulation development and quality assurance.
Sample Submission Suggestions
Detailed packaging and submission guidelines are available upon request.
Why Choose MtoZ Biolabs?
✅ Scientific Expertise: Experienced analysts and formulation scientists specializing in cellulose-based excipients.
✅ Comprehensive Methods: Integrated FTIR, GC, ICP-MS, and wet chemistry techniques for complete characterization.
✅ Quality and Compliance: Validated workflows ensuring accuracy, reproducibility, and regulatory acceptance.
✅ Flexible Collaboration: Customizable testing plans and efficient turnaround to meet diverse project needs.
✅ Global Support: Consistent, high-quality analytical services trusted by clients worldwide.
✅ One-Time-Charge: Our pricing is transparent, with no hidden fees or additional costs.
The Low-Substituted Hydroxypropyl Cellulose Analysis Service offered by MtoZ Biolabs supports pharmaceutical, nutraceutical, and material science research by ensuring excipient purity, consistency, and functionality. With advanced analytical tools and an experienced technical team, we deliver reliable data that strengthens formulation design and quality control.
MtoZ Biolabs is committed to providing professional and high-quality analytical solutions. Contact us today to learn how our services can accelerate your formulation and development success.
FAQ
Q1: How Does L-HPC Differ from Highly Substituted Hydroxypropyl Cellulose (H-HPC)?
A1: L-HPC contains 7 to 16 percent hydroxypropyl groups, giving it strong swelling capacity without full solubility. H-HPC, with higher substitution, is fully soluble and shows different viscosity and film-forming behavior.
Q2: How Is Hydroxypropyl Content Measured in L-HPC?
A2: The hydroxypropyl content is determined using gas chromatography with an internal standard or a two-step titration method involving sodium hydroxide and sodium thiosulfate. The titrant volume used corresponds to the calculated hydroxypropyl level.
How to order?







