How Does Histone Phosphorylation Influence Chromatin Structure?
-
H3S10 (serine at position 10)
-
H3T11 (threonine at position 11)
-
H2AXS139 (γ-H2AX, a hallmark marker of DNA damage)
-
Electrostatic modulation: Histones are positively charged, whereas DNA carries a negative charge. The addition of negatively charged phosphate groups reduces the overall positive charge of histones, thereby weakening histone-DNA interactions
-
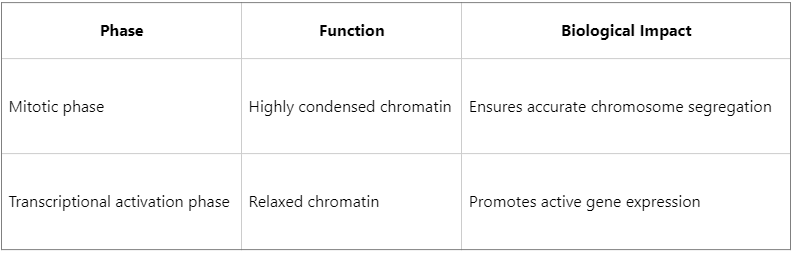
Regulation of chromatin condensation dynamics: For example, phosphorylation of H3S10 during mitotic prophase promotes chromatin condensation and ensures accurate chromosome segregation. In contrast, dephosphorylation during interphase facilitates chromatin relaxation
-
H3S10 phosphorylation synergizes with H3K9 acetylation to promote active transcription.
-
H3S10 phosphorylation can inhibit H3K9 methylation, thereby reducing heterochromatin formation.
-
Recruitment of chromatin remodeling complexes (e.g., SWI/SNF)
-
Initiation of DNA repair signaling (e.g., γ-H2AX recruits repair factors such as MDC1 and ATM)
-
Prevention of transcriptional repression complex binding
-
Aberrant gene expression
-
Chromosomal instability
-
Immune evasion of cancer cells
-
Inhibition of Aurora kinase → suppression of H3S10 phosphorylation → blockade of tumor cell division
-
Targeting the γ-H2AX signaling pathway → enhancement of radiotherapy and chemotherapy sensitivity
-
Construction of histone modification profiles
-
Quantitative analysis of site-specific phosphorylation
-
Integrated multi-omics analysis (proteomics and epigenomics)
Histone phosphorylation is an important post-translational modification that plays a critical role in the regulation of chromatin structure, gene expression, and DNA damage responses. By introducing negatively charged phosphate groups, histone phosphorylation alters the electrostatic interactions between histones and DNA. In coordination with other post-translational modifications, it contributes to the formation of the epigenetic “histone code,” thereby influencing whether chromatin adopts an open or condensed conformation. Specific phosphorylation sites, such as H3S10 and γ-H2AX, are known to play key roles in cell-cycle progression and cellular stress responses and have emerged as important molecular markers in cancer research and targeted therapeutic strategies.
Introduction to Histone Phosphorylation
Histones are the core protein components of nucleosomes and mainly include H2A, H2B, H3, and H4. The N-terminal tails of histones are highly susceptible to enzymatic modifications, including methylation, acetylation, ubiquitination, and phosphorylation.
Histone phosphorylation primarily occurs on serine (Ser), threonine (Thr), or tyrosine (Tyr) residues and is typically catalyzed by specific protein kinases.
Common phosphorylation sites include:
These modifications are closely associated with a variety of biological processes, including cell-cycle progression, DNA repair, and chromatin remodeling.
Mechanisms by Which Histone Phosphorylation Influences Chromatin Structure
1. Regulation of Chromatin Compaction
Histone phosphorylation can modulate chromatin compaction by altering the electrostatic properties of histones and weakening their affinity for DNA:
2. Acting as an Epigenetic “Code” That Influences Other Modifications
Histone phosphorylation frequently cooperates with or antagonizes other histone modifications to form the so-called histone code, collectively determining chromatin states:
This cross-regulatory mechanism enables histone phosphorylation to play an integrative role in chromatin state regulation.
3. Recruitment or Exclusion of Specific Regulatory Proteins
Certain phosphorylation sites can be specifically recognized by reader proteins, thereby mediating downstream regulatory events:
This mechanism is particularly critical during the DNA damage response, where histone phosphorylation functions as an important signaling event in cellular stress pathways.
Representative Research Case: The Dual Role of H3S10 Phosphorylation
H3S10 phosphorylation is one of the most extensively studied histone phosphorylation sites and exhibits functional duality:
Significance of Histone Phosphorylation in Disease and Therapy
1. Aberrant Phosphorylation in Cancer
Certain kinases, such as Aurora B and MSK1, are frequently overactivated in tumors, leading to abnormal histone phosphorylation and consequently promoting:
2. Development of Phosphorylation-Targeted Therapeutics
Targeting histone kinases or epigenetic regulatory proteins (readers/writers) provides promising opportunities for the development of precision epigenetic therapies. For example:
Application of Mass Spectrometry in Histone Phosphorylation Research
Comprehensive analysis of histone phosphorylation sites remains challenging using conventional methods. However, mass spectrometry technologies - particularly high-resolution LC-MS/MS - have become essential tools for investigating site specificity, dynamic changes, and modification crosstalk.
At MtoZ Biolabs, we utilize Orbitrap-based high-resolution mass spectrometry platforms combined with efficient enrichment strategies (such as TiO₂ or IMAC) and PTM database-assisted analysis to provide:
Histone phosphorylation regulates chromatin architecture, recruits regulatory proteins, and interacts with other epigenetic modifications, thereby playing multiple roles in cell-cycle regulation, DNA repair, and transcriptional control. With continuous advances in mass spectrometry technologies, our understanding of this dynamic modification continues to deepen. For researchers seeking accurate and reliable mass spectrometry data for histone modification studies, MtoZ Biolabs provides comprehensive epiproteomics solutions to support cutting-edge scientific research.
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
How to order?







