Biomarker Testing and Analysis Service
MtoZ Biolabs provides comprehensive Biomarker Testing and Analysis Service designed to support biomarker discovery, assay development, and quantitative evaluation across a wide range of therapeutic programs. By integrating multi-platform analytical technologies with expert method development and validation, we deliver high-quality biomarker data that inform mechanism studies, pharmacodynamic assessments, and translational decision making.
Overview
Biomarkers are measurable molecular indicators of physiological states, disease progression, and drug response. They may derive from proteins, peptides, metabolites, lipids, glycans, or nucleic acids and are widely used in early diagnosis, risk assessment, pharmacodynamic studies, and mechanism exploration.
Effective biomarker profiling enables researchers to:
Identify molecular alterations associated with disease onset and progression
Measure biological responses to therapeutic interventions
Support patient stratification and treatment-response prediction
Elucidate signaling pathways and molecular mechanisms
Generate data that inform translational and companion diagnostic development
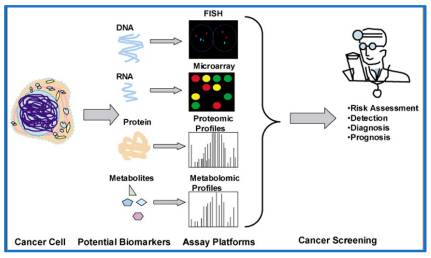
Ahmad, A. et al. PHARMACEUTICS. 2023.
Figure 1. Various Approaches for Biomarker Detection and Analysis Assays
Biomarker Testing and Analysis Service at MtoZ Biolabs
MtoZ Biolabs provides a full suite of Biomarker Testing and Analysis Services designed to accommodate exploratory studies, targeted validation, and comprehensive mechanistic investigations.
1. Custom Biomarker Assay Development
Tailored assay design and optimization for specific biomarker targets, including selection of assay format, reagents, and detection strategy.
2. Quantitative Biomarker Testing
Accurate measurement of protein, peptide, nucleic acid, cytokine, chemokine, and metabolite biomarkers using high-sensitivity detection systems.
3. Biomarker Validation and Performance Assessment
Evaluation of assay reproducibility, linearity, specificity, parallelism, dilution integrity, and biomarker stability to ensure reliable study outcomes.
4. Multiplex Biomarker Profiling
Simultaneous detection of multiple biomarkers to support pathway analysis, immune profiling, and disease signature evaluation.
5. Testing Using Commercial Panels and Kits
Implementation, verification, and performance assessment of commercially available biomarker kits for rapid and standardized analyses.
Why Choose MtoZ Biolabs?
✅ Multi-Platform Biomarker Analytics: Orbitrap-based mass spectrometers, Q Exactive HF, timsTOF Pro, ELISA, Luminex, qPCR, and additional immunoassay systems provide precise and reproducible biomarker quantification across diverse molecular classes.
✅ Integrated Multi-Omics Interpretation: Biomarker results can be combined with proteomics, metabolomics, lipidomics, and transcriptomics to deliver comprehensive biological insights and mechanism-driven conclusions.
✅ Cross-Species Analytical Capability: Biomarker testing is supported for human, non-human primate, rodent, pig, dog, and additional research species to meet diverse preclinical and translational research needs.
✅ Expert Scientific and Project Teams: Experienced specialists in disease biology, pharmacology, and toxicology provide scientific oversight and clear communication throughout every project stage.
✅ One-Time-Charge: Our pricing is transparent, no hidden fees or additional costs.
Start Your Project with MtoZ Biolabs
Advance your biomarker research with precise, reproducible, and decision-oriented analysis. MtoZ Biolabs delivers high-quality biomarker data to support discovery, pharmacodynamic evaluation, and translational development.
Contact us today to initiate your biomarker testing program.
FAQ
Q1: What types of samples are suitable?
Our Biomarker Testing and Analysis Service supports protein, peptide, cytokine, chemokine, nucleic acid, metabolite, and cell-based biomarkers across serum, plasma, CSF, tissue lysates, and other biological matrices.
Q2: What is the service general workflow?
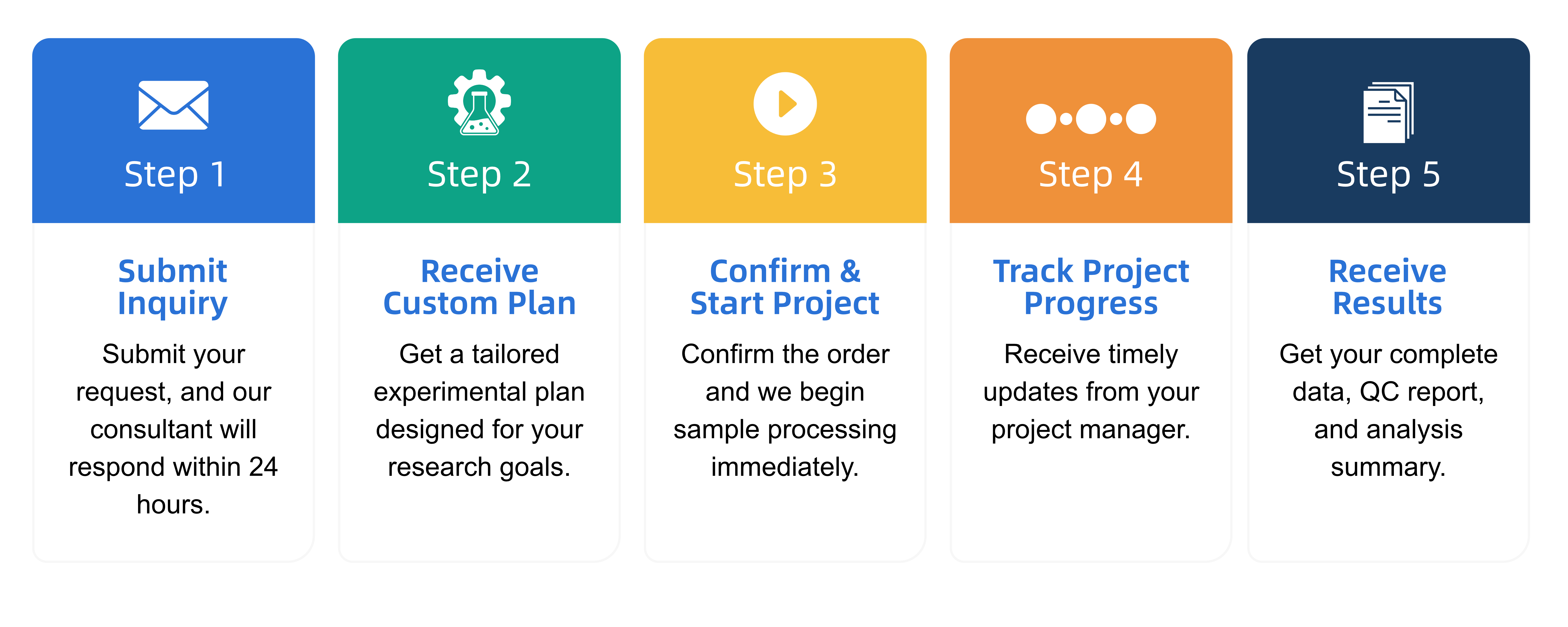
Q3: What data formats are provided?
Deliverables include raw platform files, processed datasets in Excel or CSV, graphical biomarker summaries, assay performance details, and comprehensive interpretive reports.
Q4: How should I prepare my samples?
· Recommended quantity
Generally 50–200 µL of biofluid or 20–50 mg of tissue; specific amounts vary with assay type.
· Storage and transport
Store samples at –80°C and avoid freeze-thaw cycles; ship on dry ice for stability.
· Matrix considerations
Avoid substances that may interfere with detection, including high salt concentrations, detergents, or extreme pH conditions.
For more information, please refer to Sample Submission Guidelines for Proteomics. If you are unsure about sample preparation, our team is available for consultation and can guide you through the process to ensure optimal results.
Related Services
How to order?







