What Is Histone Butyrylation (Kbu)?
-
The butyryl group is derived from butyryl-CoA.
-
The modification occurs on the ε-amino group of the lysine side chain.
-
Like acetylation (Kac), it belongs to the class of lysine acylation modifications.
-
Fatty acid β-oxidation
-
Butyrate metabolism produced by gut microbiota
-
Various amino acid metabolic pathways
-
Members of the histone deacetylase (HDAC) family
-
Sirtuins (e.g., SIRT family proteins)
-
Neutralize the positive charge of lysine residues.
-
Reduce electrostatic interactions between histones and DNA
-
Promote chromatin relaxation
-
Facilitate gene transcription
-
Elevated lipid metabolic activity
-
Enhanced gut microbial metabolism
-
Metabolic reprogramming of cellular energy pathways
-
Cancer metabolic regulation
-
Inflammatory responses
-
Neurological disorders
-
Gut microbiota-host interactions
-
Histone protein extraction
-
Proteolytic digestion
-
Enrichment using anti-Kbu specific antibodies
-
Detection by liquid chromatography-tandem mass spectrometry (LC-MS/MS)
-
Database searching and modification site identification
-
High site-specific resolution
-
Simultaneous detection of multiple lysine acylation modifications
-
Orbitrap-based mass spectrometers
-
Q-TOF systems
-
Optimized sample preparation workflows
-
Effective false discovery rate (FDR) control
-
Accurate database search parameter settings
-
Comprehensive bioinformatics analyses
-
Quantitative analysis can also be performed using Label-free or TMT-based strategies.
-
Low modification abundance
-
Small mass difference from acetylation (Kac), leading to possible misidentification
-
Antibody specificity limitations
-
Complex data processing and interpretation
-
Integrated multi-acylation modification omics
-
Single-cell level epigenetic modification analysis
-
Combined metabolomics and epigenomics studies
-
Applications in cancer biology and immune regulation
-
High-resolution Orbitrap mass spectrometry systems
-
Established acylation enrichment strategies
-
Standardized quality control frameworks
-
Advanced bioinformatics analysis capabilities
-
Histone post-translational modification proteomics
-
High-precision detection of multiple lysine acylation modifications (Kac, Kbu, Kcr, etc.)
-
Integrated quantitative proteomics and PTM-omics analysis
With the rapid advancement of epigenetics, histone post-translational modifications (PTMs) have become a central focus for understanding the mechanisms underlying gene expression regulation. From classical modifications such as acetylation (Kac) and methylation (Kme) to more recently identified acylation modifications including crotonylation (Kcr) and butyrylation (Kbu), an increasing number of novel histone acylations have been discovered. Among these, histone butyrylation (Kbu), a lysine acylation modification, has recently emerged as an important research focus at the interface between epigenetics and cellular metabolism.
What Is Histone Butyrylation (Kbu)?
Histone butyrylation (Kbu) is a post-translational modification in which a butyryl group (-CO-CH₂-CH₂-CH₃) is covalently attached to the ε-amino group of lysine (Lys, K) residues on histone proteins.
From a chemical structural perspective:
The key differences are summarized below:
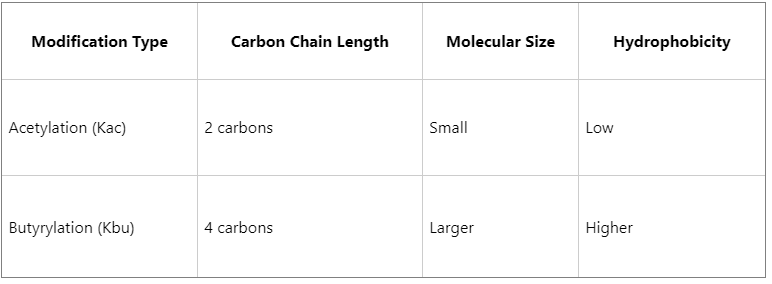
Because the butyryl group contains a longer carbon chain, Kbu may exert stronger steric and hydrophobic effects, potentially leading to more pronounced influences on chromatin structure and protein conformation.
Formation Mechanism of Kbu: A Metabolism-Driven Epigenetic Modification
1. Butyryl-CoA as the Direct Donor
The immediate donor for histone butyrylation is butyryl-CoA, which can originate from multiple metabolic pathways, including:
Therefore, Kbu represents a metabolism-coupled epigenetic modification.
2. Writer Enzymes and Eraser Enzymes
Studies have shown that certain histone acetyltransferases (HATs), such as p300/CBP, are capable of catalyzing both acetylation and butyrylation.
Potential enzymes responsible for removing butyrylation modifications may include:
These findings indicate that Kbu and Kac share partially overlapping enzymatic mechanisms. However, whether dedicated butyryltransferases exist remains an active area of investigation.
Biological Functions of Kbu
1. Regulation of Gene Transcription
Similar to histone acetylation, butyrylation can:
Previous studies indicate that Kbu is enriched in actively transcribed genomic regions, suggesting a close association with transcriptional activation.
2. Close Association with Cellular Metabolic State
Because butyryl-CoA levels are regulated by cellular metabolism, several metabolic conditions may influence Kbu levels, including:
Consequently, Kbu represents a critical molecular link connecting metabolic reprogramming, epigenetic regulation, and disease development.
3. Potential Roles in Disease Research
Preliminary studies suggest that Kbu may be involved in:
However, the precise biological functions and site-specific regulatory mechanisms of Kbu remain to be fully elucidated.
Methods for Detecting Histone Butyrylation
Because Kbu is typically present at low abundance, its detection largely relies on high-resolution mass spectrometry-based proteomics approaches.
1. Antibody Enrichment Combined with LC-MS/MS
A typical analytical workflow includes:
This strategy offers several advantages:
2. High-Resolution Mass Spectrometry Platforms
Commonly used instruments include:
High-quality datasets rely on:
Technical Challenges in Kbu Research
Despite its promising research potential, several technical challenges remain:
Therefore, comprehensive mass spectrometry strategies and well-established data analysis pipelines are essential.
Emerging Research Trends in Histone Butyrylation
Current research directions include:
In the future, Kbu is likely to become another key epigenetic regulatory marker following histone acetylation.
Importance of Professional Histone Butyrylation Analysis Services
Because Kbu detection requires stringent standards in sample preparation, mass spectrometry resolution, and bioinformatics analysis, selecting a professional technology platform equipped with the following capabilities is essential for reliable research outcomes:
MtoZ Biolabs has long specialized in:
Through optimized antibody enrichment workflows and high-resolution LC-MS/MS platforms, we enable high-coverage identification of Kbu modification sites and accurate quantitative analysis, providing robust data support for metabolism–epigenetics research.
Histone butyrylation (Kbu) serves as a key molecular bridge linking cellular metabolic states with gene expression regulation. As an emerging lysine acylation modification, it holds significant promise in studies of cancer, metabolic diseases, and microbiome-host interactions. With advances in high-resolution mass spectrometry and multi-omics integration, research on Kbu is rapidly evolving from the discovery phase toward mechanistic investigations. If you are conducting histone butyrylation or multi-acylation modification studies, selecting a robust and reliable mass spectrometry analysis platform can greatly enhance both data depth and research efficiency. MtoZ Biolabs is committed to providing professional support for your epigenetics research.
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
How to order?







