Guide to Antibody Characterization: From Sample Preparation to Data Interpretation
-
Confirm antibody sequences and molecular architecture.
-
Establish binding activity and specificity.
-
Evaluate stability, purity, and biological activity.
-
LC-MS/MS-based sequencing: Applicable to antibodies that have already been produced but lack original sequence information; enables sequence inference at the protein level.
-
cDNA sequencing: Applicable to hybridoma cells or expression vectors; directly yields antibody gene sequences at the nucleic-acid level.
-
SDS-PAGE/Western blot: A coarse assessment of apparent molecular weight and heavy-chain/light-chain profiles.
-
SEC-HPLC (size-exclusion chromatography): Separates monomers from aggregates and provides an accurate evaluation of aggregation levels.
-
CE-SDS: A high-resolution, electrophoresis-based method enabling automated assessment of heavy-chain and light-chain integrity.
-
Mass spectrometry: Detects subtle degradation and/or modifications; particularly well suited for quality-control applications.
-
MS-based glycan profiling (Glycan Profiling): Rapid identification of glycan types and their relative abundances.
-
LC-MS protein/peptide mapping: Identification of PTM sites and PTM types.
-
Capillary isoelectric focusing (cIEF): Assessment of isoelectric point shifts that indirectly reflect modification status.
-
ELISA: Rapid screening of binding to the target antigen.
-
Western blot / IHC: Verification of specific recognition across different sample types.
-
Surface plasmon resonance (SPR) / bio-layer interferometry (BLI): High-throughput and highly sensitive measurement of antigen–antibody interaction kinetics (ka, kd, KD, etc.).
-
Differential scanning calorimetry (DSC): Determination of the thermal denaturation temperature (Tm) to reflect thermal stability.
-
Dynamic light scattering (DLS): Detection of aggregation propensity in solution.
-
Accelerated stability studies: Evaluation of stability under different pH, temperature, repeated freeze-thaw cycles, and related conditions.
-
High-resolution Orbitrap and Q-TOF mass spectrometry platforms.
-
Comprehensive full-sequence antibody analysis and panoramic profiling of PTMs.
-
SPR/BLI-based affinity kinetics evaluation services.
As essential biomolecules in basic research and biomedicine, antibody performance characteristics directly determine the reliability of experimental outcomes and the effectiveness of downstream applications. Systematic antibody characterization enables the delineation of structural attributes, functional activity, and stability, and constitutes a critical step for ensuring batch-to-batch quality consistency and well-controlled functional properties, particularly in drug development, diagnostic reagents, and precision medicine.
Why Is Antibody Characterization Important?
Antibodies are highly functional and highly specific immune molecules; however, in practical use, their performance may vary substantially due to structural heterogeneity and/or differences in production and preparation methods. The central objectives of antibody characterization are to:
These efforts provide a foundational safeguard for reproducibility and credibility in basic research, and they also represent a key quality gate prior to clinical entry in areas such as antibody therapeutics development and companion diagnostics (CDx).
Core Workflow and Technical Pathways of Antibody Characterization
Antibody characterization typically comprises the following core steps, each supported by corresponding analytical technologies and interpretive readouts.
1. Antibody Sequence Confirmation (Sequence Confirmation)
(1) Purpose: To confirm sequence information for the antibody variable regions (VH, VL) and constant region (CH), thereby ensuring antibody consistency and controllability.
(2) Common Approaches
2. Antibody Purity and Integrity Analysis (Purity & Integrity)
(1) Purpose: To ensure the absence of aggregation, degradation, or undesired modifications, thereby maintaining structural stability and consistency across downstream experiments.
(2) Common Approaches
3. Antibody Glycosylation and Modification Detection (Glycosylation & PTM Analysis)
(1) Purpose: Antibody glycosylation patterns can markedly influence structural stability and Fc-mediated immune functions. Other post-translational modifications (e.g., oxidation and deamidation) should also be monitored.
(2) Common Approaches
4. Binding Activity and Affinity Evaluation (Binding Specificity & Affinity)
(1) Purpose: To verify target-specific recognition and to further quantify affinity parameters (e.g., KD).
(2) Common Approaches
5. Antibody Stability and Thermodynamic Analysis (Stability & Thermodynamics)
(1) Purpose: Physical stability during storage, transport, and use can materially affect antibody performance.
(2) Common Approaches
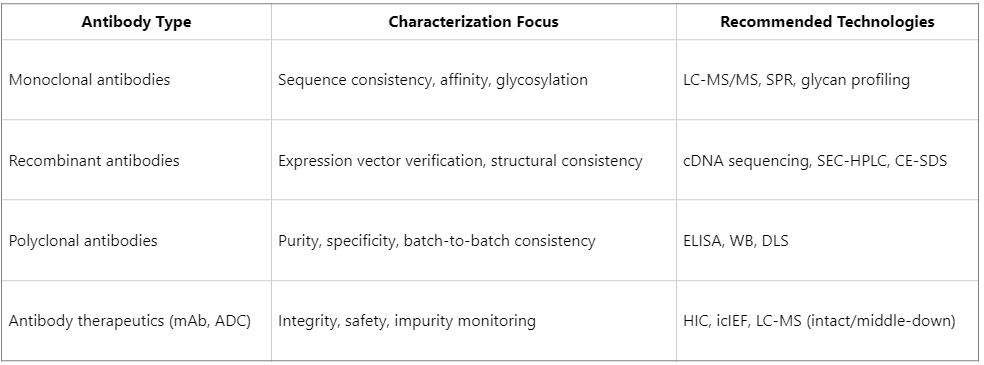
Differences in Characterization Strategy for Specific Antibody Types
Common Challenges and Optimization Suggestions in Antibody Characterization
1. Antibody Aggregation Is Difficult to Resolve
It is recommended to combine SEC-HPLC and DLS to confirm monomer content from complementary analytical dimensions.
2. Post-Translational Modifications Are Difficult to Identify
Proteomics-grade LC-MS/MS can be adopted to improve the detection rate of modifications.
3. Quality Control Is Challenging for Commercial Antibodies with Unknown Sequences
De novo MS-based sequencing combined with recombinant expression is recommended to establish a closed-loop quality verification workflow.
MtoZ Biolabs: A One-Stop Antibody Characterization Service Platform
At each critical node of antibody characterization, MtoZ Biolabs can provide customized solutions:
Whether you are conducting basic research, developing diagnostic products, or advancing an IND application for antibody therapeutics, we can help you build a scientific and reliable antibody characterization data framework.
Antibody characterization should not be treated merely as a verification step; rather, it is an essential upstream process that supports innovation and quality assurance. Through a systematic, comprehensive, and data-driven antibody characterization workflow, researchers and biopharmaceutical organizations can proceed with greater confidence in downstream R&D and clinical translation. MtoZ Biolabs remains committed to serving as a trusted antibody analytics partner, supporting high-quality research and the advancement of precision medicine.
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
How to order?







