Wax Ester Analysis Service
Wax esters are composed of a fatty alcohol molecule and a fatty acid. Organic acids usually contain a carboxyl group (-COOH), and alcohols have a hydroxyl group (-OH). When an organic acid combines with an alcohol, an ester is formed. In wax esters, the hydroxyl group of the fatty alcohol combines with the carboxyl group of the fatty acid to form an ester bond. There are various types of wax esters, mainly classified as saturated wax esters and unsaturated wax esters. Saturated wax esters have higher melting points and are generally solid at room temperature. Compared to saturated wax esters, unsaturated wax esters have lower melting points and tend to be liquid at room temperature. The carbon-chain lengths of fatty acids and fatty alcohols can vary, and there are many different combinations of fatty acids and fatty alcohols, each combination having a unique set of properties in terms of phase transition and spatial orientation.
The chain lengths of fatty acids and fatty alcohols in natural wax esters vary. In plant-derived wax esters, the fatty acids typically range between C12-C24, while the alcohols in plant wax esters can be very long, typically C24-C34. Wax esters are commonly part of leaves, shells, and the cuticle of arthropod exoskeletons, helping to prevent water loss. Plants such as beeswax and jojoba can store numerous wax esters. Marine organisms such as pelagic invertebrates, dinoflagellates, and fish utilize low-density wax esters in their swim bladders or other tissues to provide buoyancy.
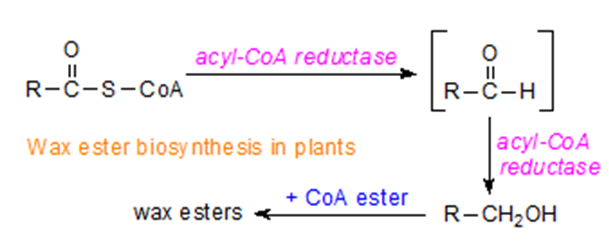
Figure 1. Wax Ester Analysis
MtoZ Biolabs offers reliable, fast, and cost-effective wax ester analysis services based on highly stable, reproducible, and sensitive systems for separation, characterization, identification, and quantitative analysis of wax ester, combined with LC-MS/MS.
How to order?







