Biopharmaceutical Disulfide Bond Analysis Service
Disulfide bonds, formed through the oxidation of two cysteine residues' sulfur atoms, are critical to the functionality and stability of protein pharmaceuticals. In biopharmaceuticals, particularly in monoclonal antibody development, correct disulfide bond pairing and location critically influence the drug's bioactivity and safety. Incorrect pairings can deactivate the drug or provoke adverse immune responses. Consequently, precise analysis and verification of disulfide bonds' conditions and positions are vital for maintaining biopharmaceutical quality and safety.
MtoZ Biolabs' biopharmaceutical disulfide bond analysis service employing high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) alongside advanced bioinformatics, MtoZ Biolabs offers extensive qualitative and quantitative disulfide bond analyses. Our methodologies precisely pinpoint all disulfide bonds within proteins, identifying both correctly formed and mismatched bonds as well as any potential free sulfhydryl groups.
Analysis Workflow
1. Sample Preparation
Proteins are treated with advanced alkylation methods to protect thiol groups and prevent erroneous disulfide bonds.
2. Enzymatic Digestion
Targeted enzymatic treatments (e.g., trypsin, pepsin) optimize peptide generation.
3. LC-MS/MS Identification
This step accurately maps all disulfide bond locations within the peptides.
4. Bioinformatics Analysis
Comprehensive tools, including pLink-SS and SlinkS, deeply analyze disulfide and free cysteine bonds.
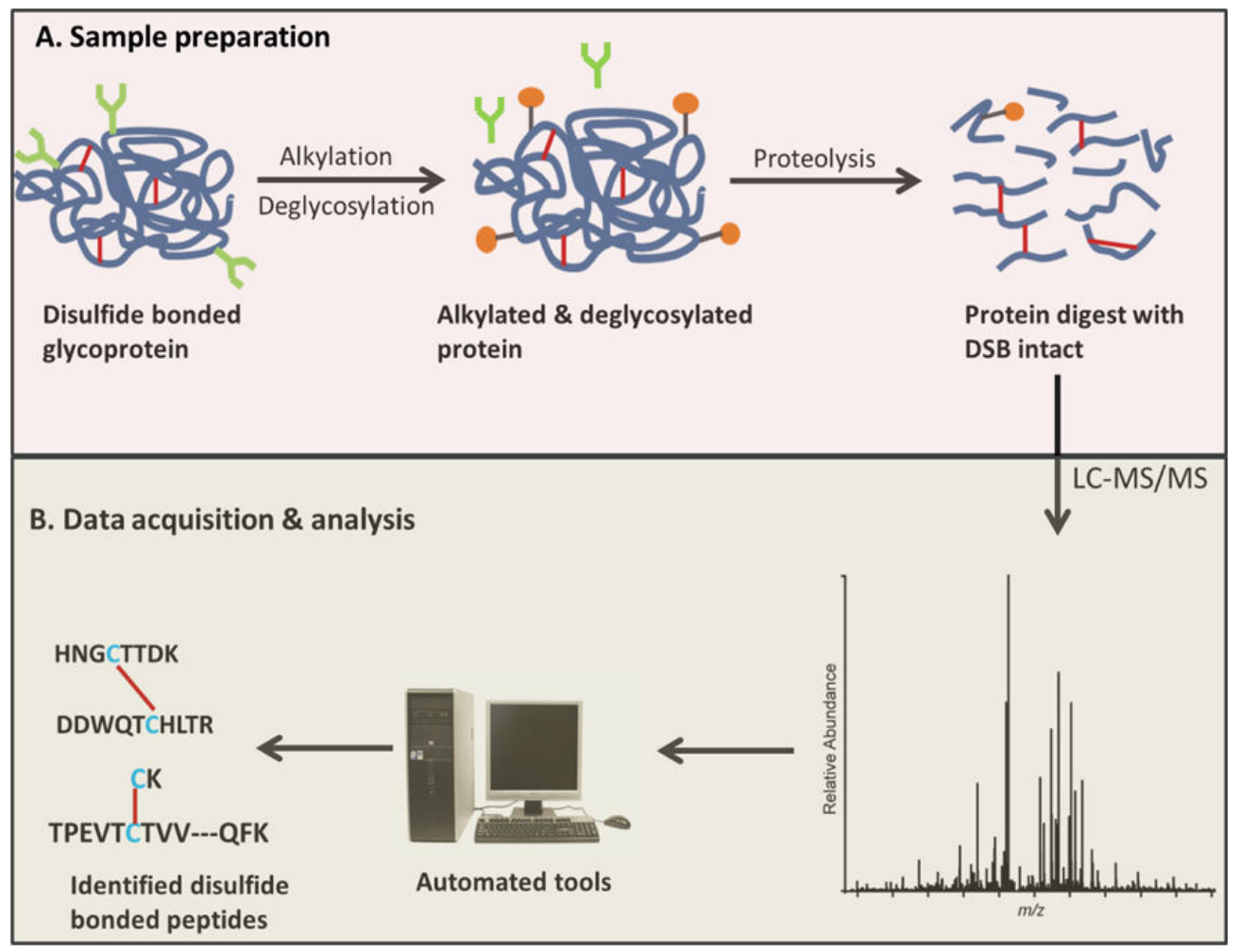
Lakbub, J. C. et al. Anal. Bioanal. Chem. 2017.
Figure 1. Disulfide Bond Analysis Workflow
Service Advantages
Precision and Expertise: Our use of cutting-edge technology ensures highly accurate, reliable analyses.
Efficiency: The analysis process is streamlined, from sample intake to report delivery, supporting rapid research and development cycles.
Customization: We tailor solutions and support to meet diverse customer needs.
Applications
Drug Development: Ensuring drug efficacy and safety by optimizing protein structures.
Quality Assurance: Regular testing of disulfide bonds to maintain drug production quality.
Preclinical Assessment: Detailed analyses aid in evaluating drug safety and effectiveness.
Regulatory Compliance: Ensuring drugs meet structural integrity standards demanded by regulators.
Deliverables
1. Experimental Procedures
2. Relevant Mass Spectrometry Parameters
3. Detailed Information on Disulfide Bonds
4. Mass Spectrometry Images
5. Raw Data
At MtoZ Biolabs, we are dedicated to delivering the highest standards of professionalism and reliability in biopharmaceutical disulfide bond analysis, advancing your research and production capabilities. For more details or to collaborate, please reach out to our team of experts.
MtoZ Biolabs, an integrated chromatography and mass spectrometry (MS) services provider.
Related Services
How to order?







