Disulfide Bond Mapping Analysis Service
Disulfide bonds are a crucial structural element in proteins, formed by the sulfur atoms from cysteine amino acid residues under oxidative conditions. These bonds play a pivotal role in the biological activity and functionality of proteins. Found extensively in proteins such as cellular surface receptors, antibodies, growth factors, hormones, and enzymes, disulfide bonds are prone to reduction or mispairing, impacting the structure and biological activity of the originating peptides or proteins. Disulfide bond localization analysis involves identifying the positions and connectivity of disulfide bonds within protein structures, enhancing our understanding of protein structure and function. In biopharmaceutical development, incorrect disulfide bond pairings can lead to drug ineffectiveness and pose potential safety risks. Thus, precise detection and management of disulfide bonds are essential for biopharmaceutical characterization.
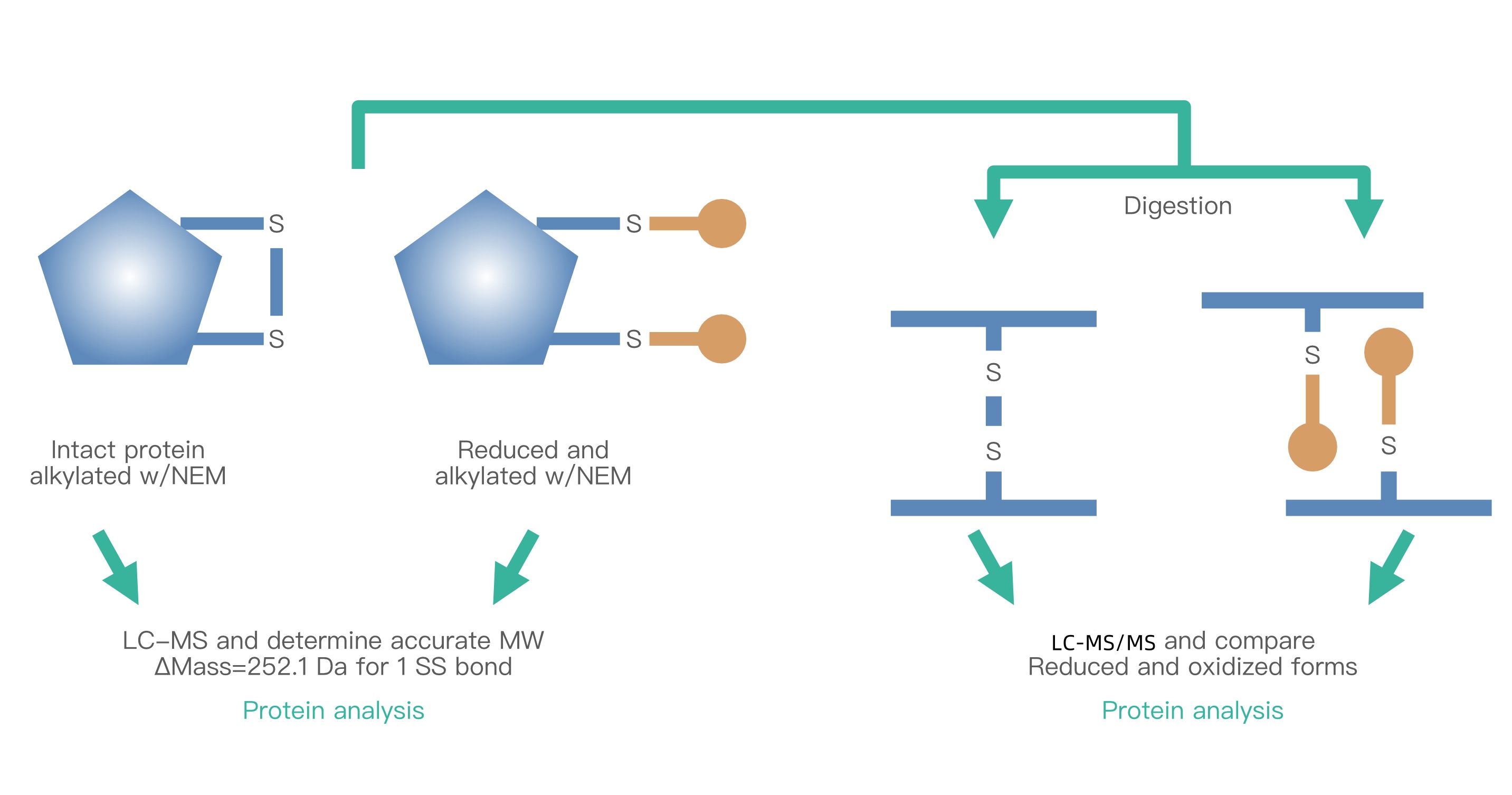
Figure 1. Principle of Disulfide Bond Localization Analysis
Advancements in LC-MS technology have significantly enhanced the sensitivity and accuracy of detection, propelling the field of disulfide bond analysis. MtoZ Biolabs utilizes a disulfide bond analysis platform that integrates HPLC and high-resolution MS, ensuring accurate and efficient identification of various disulfide bond types, including intra- and inter-chain, single and multiple, overlapping, and mismatched bonds.
Experimental Instruments
1. HPLC: Easy-nLC 1200 (Thermo Fisher Scientific)
2. High-Resolution MS: Q Exactive™ Hybrid Quadrupole-Orbitrap™ Mass Spectrometer (Thermo Fisher Scientific)
FAQ
1. What are the Advantages of MS in Disulfide Bond Identification?
MS excels in disulfide bond analysis by not only determining their presence and quantity but also their specific locations and relative concentrations. Its high sensitivity and resolution enable precise detection and quantification, even at low concentrations, making it invaluable for analysis of limited or valuable samples.
2. Why is Alkylation Performed?
Alkylation seals free thiols to prevent the mispairing of disulfide bonds during experiments. By substituting thiols with alkylthiol, alkylation prevents undesirable reactions between thiols, ensuring accurate and reliable disulfide bond information in MS, critical for understanding protein structure and function.
Deliverables
In the technical report, MtoZ Biolabs will provide you with detailed technical information, including:
1. Experimental Procedures
2. Relevant Mass Spectrometry Parameters
3. Detailed Analysis of Disulfide Bond Localization
4. Mass Spectrometry Images
5. Raw Data
How to order?







