Active Substances Screening Service
Drug development is a long and complex process, broadly divided into four main stages:
1. Target Selection and Validation
2. Compound Screening and Lead Optimization
3. Preclinical Studies
4. Clinical Trials
Drug screening refers to the evaluated process of substances (samples) potentially used as drugs, assessing their biological activity, pharmacological effects, and therapeutic value through appropriate methods. This process encompasses biochemical and cellular levels of screening, divided into high-throughput screening (HTS) and virtual screening.
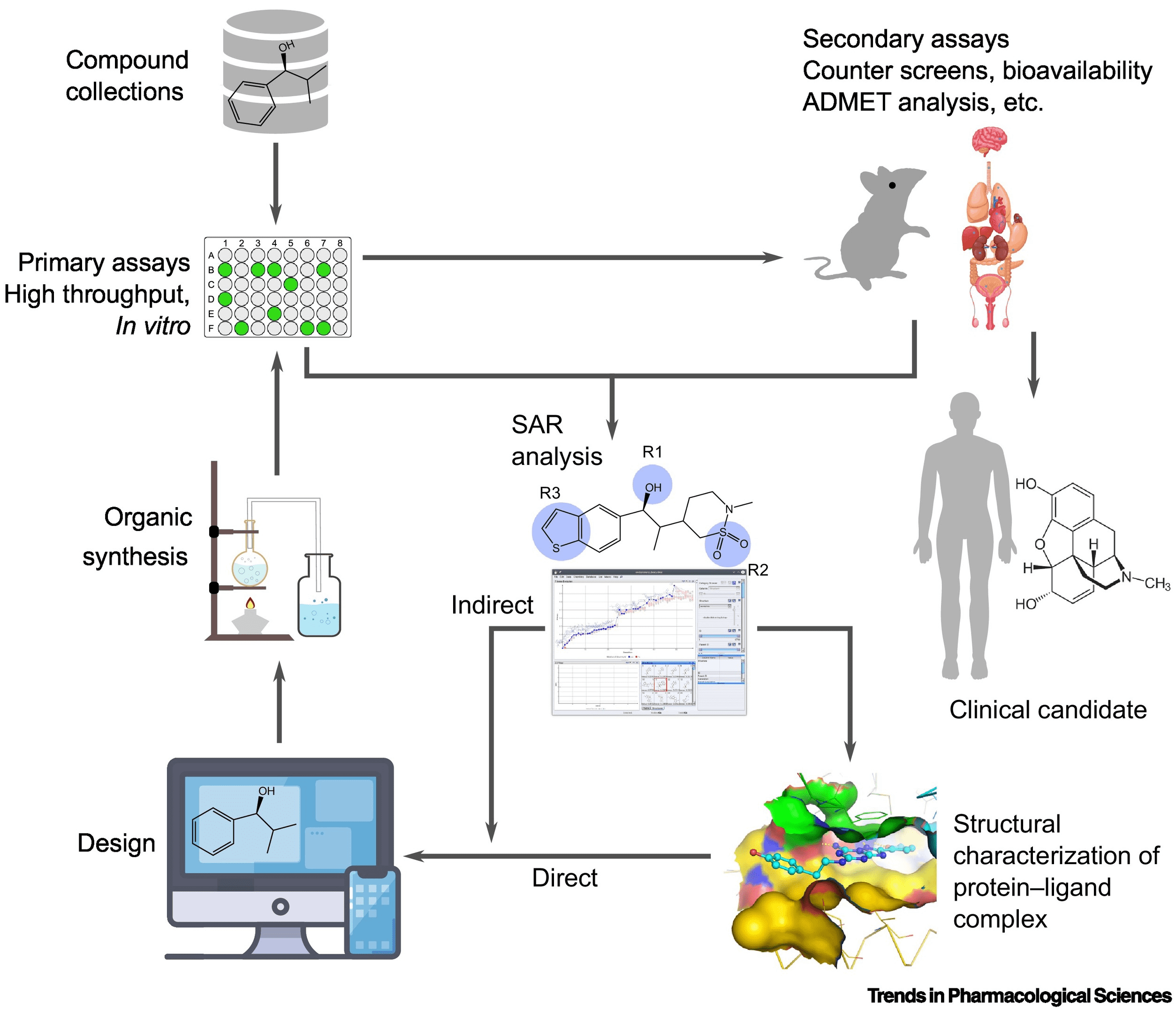
Figure 1. A Representative Drug Development Process [1]
HTS is a standardized and quantifiable experimental protocol employed in drug screening. Depending on the experimental model, HTS can be categorized into biochemical and cellular level screenings. Biochemical screenings involve designing experiments on the drugs' target, typically proteins with specific physiological functions, such as enzymes and receptors. Following the mixing of the candidate compound with the target, interactions can be quantitatively assessed using methods like enzyme-linked immunosorbent assay (ELISA), affinity mass spectrometry (ASMS), nuclear magnetic resonance (NMR), and surface plasmon resonance (SPR), which serve as the foundation for screening compounds. Cellular level screening, a model that more close to physiological conditions, targets the cells designated for drug action. Utilizing cell culture techniques to acquire the necessary cells and facilitating their interaction with candidate compounds allows for the evaluation of the compound's efficacy through detection methodologies analogous to those employed in biochemical screening.
Virtual screening represents a novel trajectory in the evolution of drug screening techniques. Given that physical drug screening necessitates the establishment of a comprehensive compound library, the extraction or cultivation of a significant number of target proteins or cells, and the support of sophisticated equipment, virtual screening emerges as a computational strategy rooted in computer-aided drug design. It enables the identification of lead or pioneer compounds within a virtual small molecule compound library, markedly reducing the costs associated with drug development. Molecular docking, a mature method based on the development of computer, is extensively applied in the virtual docking. It facilitates the identification of novel compounds of therapeutic interest, predicting ligand-target interactions at the molecular level, or elucidating structure-activity relationships (SAR) without necessitating prior knowledge of the chemical structures of other target modulators.
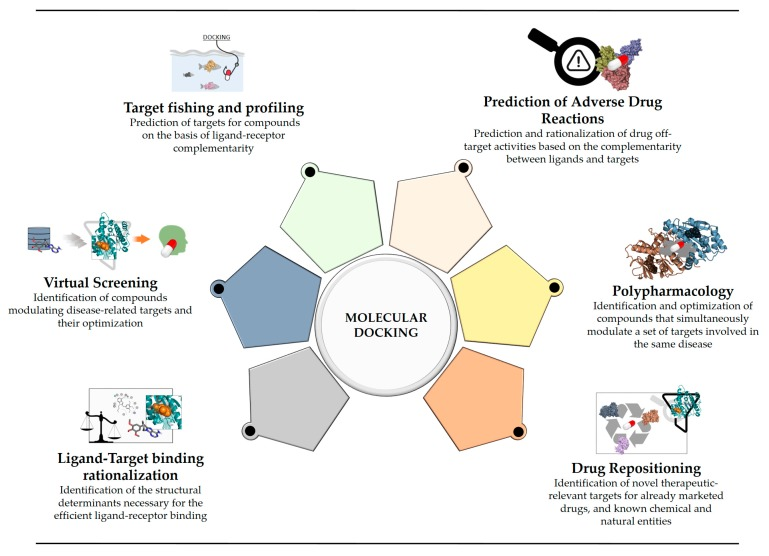
Figure 2. The Application of Molecular Docking in Drug Development [2]
MtoZ Biolabs offers a comprehensive preclinical drug development platform, providing services for the screening and identification of drug active ingredients. The team is equipped to identify the most efficacious active ingredients from a diversity of sources, through a suite of screening technologies such as ASMS, SPR, and intact protein mass spectrometry (Intact-MS). Services extend to the provision of compound libraries, pharmacological screening models, and the analysis of screening experimental data, delivering a one-stop solution for the identification and screening of active substances.
References
[1] Chan HCS, Shan H, Dahoun T, Vogel H, Yuan S. Advancing Drug Discovery via Artificial Intelligence. Trends Pharmacol Sci. 2019 Aug;40(8):592-604. doi: 10.1016/j.tips.2019.06.004. Epub 2019 Jul 15. Erratum in: Trends Pharmacol Sci. 2019 Oct;40(10):801. PMID: 31320117.
[2] Pinzi L, Rastelli G. Molecular Docking: Shifting Paradigms in Drug Discovery. Int J Mol Sci. 2019 Sep 4;20(18):4331. doi: 10.3390/ijms20184331. PMID: 31487867; PMCID: PMC6769923.
How to order?







